
Concept explainers
Draw the products of each reaction. Use the
a.
b. 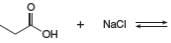
c. 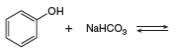
d.
(a)
Interpretation: The products of the given reaction are to be drawn. If the equilibrium favors the starting materials or a product is to be predicted.
Concept introduction: According to Bronsted-Lowry theory, when an acid donates a proton the species formed is known as conjugate base and when the base accepts a proton the species formed is known as conjugate acid.
In a reaction which strongly favors the formation of products, the base used to remove proton from the acid should be stronger than the base formed when the proton is removed. In a reaction which favors the products, equilibrium will favors the formation of the weaker acid or weaker base. The
Answer to Problem 2.49P
The products of the given reaction are
Explanation of Solution
The complete reaction between methyl amine and
The
The products of the given reaction are
(b)
Interpretation: The products of the given reaction are to be drawn. If the equilibrium favors the starting materials or a product is to be predicted.
Concept introduction: According to Bronsted-Lowry theory, when an acid donates a proton the species formed is known as conjugate base and when the base accepts a proton the species formed is known as conjugate acid.
In a reaction which strongly favors the formation of products, the base used to remove proton from the acid should be stronger than the base formed when the proton is removed. In a reaction which favors the products, equilibrium will favors the formation of the weaker acid or weaker base. The
Answer to Problem 2.49P
The products of the given reaction are
Explanation of Solution
The complete reaction between propanoic acid and sodium chloride is shown below.
The
The products of the given reaction are
(c)
Interpretation: The products of the given reaction are to be drawn. If the equilibrium favors the starting materials or a product is to be predicted.
Concept introduction: According to Bronsted-Lowry theory, when an acid donates a proton the species formed is known as conjugate base and when the base accepts a proton the species formed is known as conjugate acid.
In a reaction which strongly favors the formation of products, the base used to remove proton from the acid should be stronger than the base formed when the proton is removed. In a reaction which favors the products, equilibrium will favors the formation of the weaker acid or weaker base. Or the
Answer to Problem 2.49P
The products of the given reaction are
Explanation of Solution
The complete reaction between phenol and sodium hydrogen carbonate is shown below.
The
The products of the given reaction are
(d)
Interpretation: The products of the given reaction are to be drawn. If the equilibrium favors the starting materials or a product is to be predicted.
Concept introduction: According to Bronsted-Lowry theory, when an acid donates a proton the species formed is known as conjugate base and when the base accepts a proton the species formed is known as conjugate acid.
In a reaction which strongly favors the formation of products, the base used to remove proton from the acid should be stronger than the base formed when the proton is removed. In a reaction which favors the products, equilibrium will favors the formation of the weaker acid or weaker base. Or the
Answer to Problem 2.49P
The products of the given reaction are
Explanation of Solution
The complete reaction between ethyne and ethyl lithium is shown below.
The
The products of the given reaction are
Want to see more full solutions like this?
Chapter 2 Solutions
Organic Chemistry
Additional Science Textbook Solutions
Organic Chemistry
Chemistry: The Molecular Nature of Matter
Chemistry: Structure and Properties (2nd Edition)
Thermodynamics, Statistical Thermodynamics, & Kinetics
Chemistry
Living By Chemistry: First Edition Textbook
- Give the major product of the following reaction. HBr Ph-C C-Ph НООН There is no reaction under these conditions or the correct product is not listed here. Br Ph Ph Br Br Ph Ph Br Br Ph Ph Br Br Ph Ph Br Br J Ph Ph Br Br Ph Ph Brarrow_forwardWhat are the equilibrium concentrations of acetic acid, the acetate ion, and H3O+ for a 0.10 M solution of acetic acid (K2 = 1.8 105)? What is the pH of the solution?arrow_forwardWhat are the products of each of the following acid-base reactions? Indicate the acid and its conjugate base and the base and its conjugate acid. HC1O4+ H2O — NH/ + H2O -» HCOr + OH" —arrow_forward
- The Keq for the reaction: A + B ↔ AB is 2.163 What is the Keq for 2 + AB ↔ 2 A + 2 Barrow_forwardBased on the following equilibrium H++ HCO3- ↔ H2CO3 A)State what would happen if we added HCl to the reaction B) State what would happen if we added NaOH to the reaction.arrow_forwardDisulfides are compounds that have S¬ S bonds, like peroxides have O¬ O bonds. Thiols are organic compounds that have the general formula R¬ SH, where R is a generic hydrocarbon. The SH- ion is the sulfur counterpart of hydroxide, OH-. Two thiols can react to make a disulfide, R¬ S¬ S¬ R. If you wanted to convert a disulfide to two thiols, should you add a reducing agent or oxidizing agent to the solution?arrow_forward
- Acid Base Reaction Consider the following acid-base reaction. HSO4 + CN-HCN + SO4²- Which compound is the conjugate base of the Bronsted-Lowry acid? HCN HSO4 CN SO4²- 0 0 0 0 ξ0000 Which compound is the conjugate acid of the Bronsted-Lowry base? CN SO4²- HSO4 HCN Darrow_forwardWill the following reaction occur? O NH O Yes O No EtOH cat. H₂SO4 H₂N O OEtarrow_forwardThe conjugate base of benzoic acid is used as a 72 preservative. Write the equilibrium reaction for this weak acid in aqueous solution. ОН Benzoic acid a. What substances are present at equilibrium? b. At equilibrium, are reactants or products favored, and why? c. Are the concentrations of benzoic acid and benzoate constant or changing at equilibrium? d. What happens to the equilibrium if more hydronium ions are added to the reaction? e. Label the conjugate acid-base pairs.arrow_forward
- 18). The formulas and the structures of two acids are shown below. Which acid is a stronger acid? If the acid strength of the two acids is equal, say so. Cl2CHCOOH CH3COOH но но CI-C-C-OH н-с-с-он ČI Ką = 0.0324 Ką = 1.82 x 10-5 %3D a. Cl¿CHCOOH is a stronger acid than CH;COOH. b. CH;COOH is a stronger acid than CI½CHCOOH c. The two have equal acid strength. d. none of the abovearrow_forwardWhich ligand is most likely to stabilize a metal in a low oxidation state? O H2O NO O NH3arrow_forwardWhat is the structure of the conjugate acid for the reaction shown below? NH OK NH3 C OA Oc D 00arrow_forward
 Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning





