
Concept explainers
(a)
Interpretation:
The isomeric structures of octane having five carbons in their principal chains are to be drawn. The naming for all the structures is to be stated.
Concept introduction:
The naming of the chemical compound is done using the parameters given by IUPAC. IUPAC stands for International Union of Pure and Applied Chemistry. This system of nomenclature is accepted worldwide. The IUPAC system provides the set of rules in order to do the naming of the chemical compounds.
Isomers are the chemical compounds which have same chemical formula but different arrangement of molecules.
Answer to Problem 2.26AP
The isomeric structures of octane having five carbons in their principal chains along with their names are drawn below as,
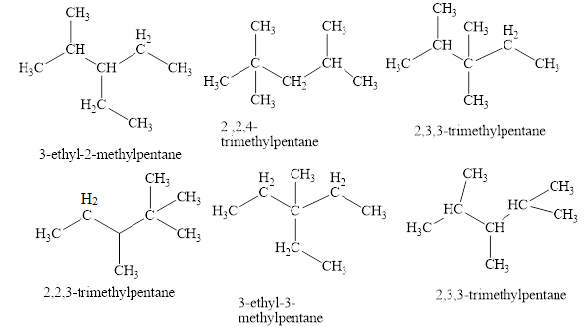
Explanation of Solution
The structure of octane can be drawn as shown in figure 1.
![]()
Figure 1
The isomers of the octane having five carbons in their principal chains can be drawn by rearranging the carbon atoms.
The first isomer is shown in figure 2 as,
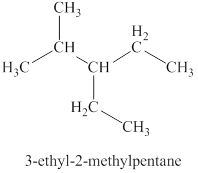
Figure 2
For the naming of the compound, the longest carbon chain is selected first. In the given compound the longest carbon chain has
The second isomer is shown in figure 3 as,
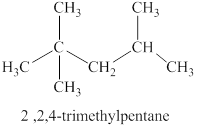
Figure 3
For the naming of the compound the longest carbon chain is selected first. In the given compound the longest carbon chain has
The third isomer is shown in figure 4 as,
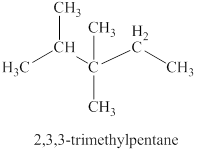
Figure 4
For the naming of the compound the longest carbon chain is selected first. In the given compound the longest carbon chain has
The fourth isomer is shown in figure 5 as,
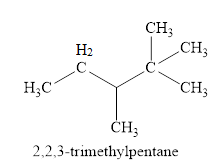
Figure 5
For the naming of the compound the longest carbon chain is selected first. In the given compound the longest carbon chain has
The fifth isomer is shown in figure 6 as,
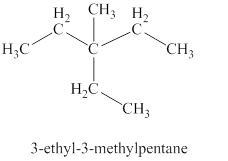
Figure 6
For the naming of the compound the longest carbon chain is selected first. In the given compound the longest carbon chain has
The sixth isomer is shown in figure 7 as,
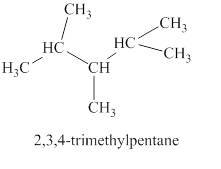
Figure 7
For the naming of the compound the longest carbon chain is selected first. In the given compound the longest carbon chain has
The isomeric structures of octane having five carbons in their principal chains along with their names are shown in figure 2 to 7.
(b)
Interpretation:
The isomeric structures of octane having six carbons in their principal chains are to be drawn. The naming for all the structures is to be stated.
Concept introduction:
The naming of the chemical compound is done using the parameters given by IUPAC. IUPAC stands for International Union of Pure and Applied Chemistry. This system of nomenclature is accepted worldwide. The IUPAC system provides the set of rules in order to do the naming of the chemical compounds.
Isomers are the chemical compounds which have same chemical formula but different arrangement of molecules.
Answer to Problem 2.26AP
The isomeric structures of octane having six carbons in their principal chains along with their names are drawn below as,
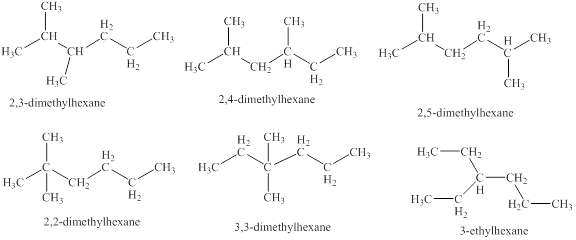
Explanation of Solution
The isomers of the octane having six carbons in their principal chains can be drawn by rearranging the carbon atoms.
The first isomer is shown in figure 8 as,
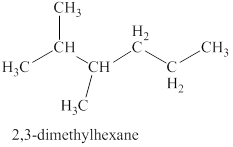
Figure 8
For the naming of the compound the longest carbon chain is selected first. In the given compound the longest carbon chain has
The second isomer is shown in figure 9 as,
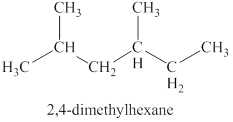
Figure 9
For the naming of the compound the longest carbon chain is selected first. In the given compound the longest carbon chain has
The third isomer is shown in figure 10 as,
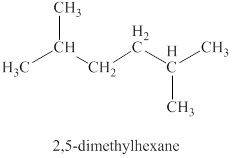
Figure 10
For the naming of the compound the longest carbon chain is selected first. In the given compound the longest carbon chain has
The fourth isomer is shown in figure 11 as,
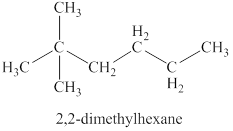
Figure 11
For the naming of the compound the longest carbon chain is selected first. In the given compound the longest carbon chain has
The fifth isomer is shown in figure 12 as,
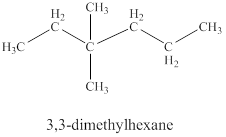
Figure 12
For the naming of the compound the longest carbon chain is selected first. In the given compound the longest carbon chain has
The sixth isomer is shown in figure 13 as,
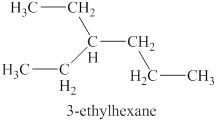
Figure 13
For the naming of the compound the longest carbon chain is selected first. In the given compound the longest chain carbon has
The isomeric structures of octane having six carbons in their principal chains along with their names are shown in figure 8 to 13.
Want to see more full solutions like this?
Chapter 2 Solutions
Organic Chemistry
- (a) Write a chemical equation involving structural formulas for the catalytic cracking of decane into an alkaneand an alkene that contain equal numbers of carbonatoms. Assume that both products have straight chainsof carbon atoms.(b) Draw and name one other isomer of the alkene.arrow_forward(a) What structural feature is associated with each type of hydrocarbon: alkane, cycloalkane, alkene, and alkyne?(b) Give the general formula for each type.(c) Which hydrocarbons are considered saturated?arrow_forward(a) What is the molecular formula of hexane, the alkane withsix carbonsarrow_forward
- Cyclopropane (C3H6, a three-membered ring) is more reactive than most other cycloalkanes.(a) Draw a Lewis structure for cyclopropane.(b) Compare the bond angles of the carbon atoms in cyclopropane with those in an acyclic (noncyclic) alkane.(c) Suggest why cyclopropane is so reactive.arrow_forwardWrite the chemical formula and Lewis structure of the following, each of which contains five carbon atoms:(a) an alkane(b) an alkene(c) an alkynearrow_forward(a) Draw the condensed structural formulas for the cis andtrans isomers of 2-pentene. (b) Can cyclopentene exhibitcis–trans isomerism? Explain. (c) Does 1-pentyne haveenantiomers? Explain.arrow_forward
- Indicate whether each statement is true or false. (a) Pentanehas a higher molar mass than hexane. (b) The longer the linearalkyl chain for straight-chain hydrocarbons, the higherthe boiling point. (c) The local geometry around the alkynegroup is linear. (d) Propane has two structural isomers.arrow_forwardGive the molecular formula of a hydrocarbon containingfive carbon atoms that is (a) an alkane, (b) a cycloalkane,(c) an alkene, (d) an alkyne.arrow_forwardIndicate whether each statement is true or false. (a) Twogeometric isomers of pentane are n-pentane and neopentane.(b) Alkenes can have cis and trans isomers around theCC double bond. (c) Alkynes can have cis and trans isomersaround the CC triple bond.arrow_forward
- (a) The compound given below had the following IUPAC name and structural formula dibromocyclopentane C3H6CHBrCHBr (i) What type of isomerism is possible in the organic compound? (ii) Draw all the pairs of possible isomers and name them.arrow_forwardGive the molecular formula of a hydrocarbon containingsix carbon atoms that is (a) a cyclic alkane, (b) a cyclicalkene, (c) a linear alkyne, (d) an aromatic hydrocarbon.arrow_forwardFor each of the following, give an example of a member compound containing 5 carbons total: (a) Alcohol (b) Amine(c) Carboxylic acid (d) Etherarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





