
Concept explainers
a)
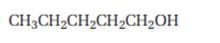
Interpretation:
Starting from 1-pentanol how pentanal can be prepared is to be shown.
Concept introduction:
Primary alcohols are oxidized to carboxylic acids by strong oxidizing agents like CrO3 and KMnO4 etc. The oxidation can be stopped in the
To show:
How to prepare pentanal starting from 1-pentanol.
b)
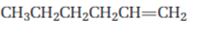
Interpretation:
Starting from 1-hexene how pentanal can be prepared is to be shown.
Concept introduction:
When treated with ozone,
To show:
How to prepare pentanal starting from 1-hexene.
c)
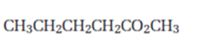
Interpretation:
Starting from methylpentanoate how pentanal can be prepared is to be shown.
Concept introduction:
To show:
How to prepare pentanal starting from methylpentanoate.
d)
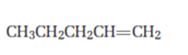
Interpretation:
Starting from 1-pentene how pentanal can be prepared is to be shown.
Concept introduction:
Alkenes when subjected to hydroboration-oxidation yield primary alcohols which when oxidized with Dess-Martin periodinate in dichloromethane yield aldehydes.
To show:
How to prepare pentanal starting from 1-pentene.
Trending nowThis is a popular solution!

Chapter 19 Solutions
Organic Chemistry
- (a) Draw the structure of the hemiacetal formed from one mole of benzaldehyde and one mole of ethanol. (b) Draw the structure of the acetal formed from one mole of benzaldehyde and two moles of ethanol. (c) Draw the structure of 2-methoxy-2-butanol. What compounds could you prepare this from? (d) Draw the structure of 3-methoxyl-2-butanol. What functional groups are present? Is this an acetal, a hemiacetal, or neither? Explain. (e) Identify the functional groups in the molecules shown below. Circle any acetals or hemiacetal, and identify which they are. 0-arrow_forwardComparing Two Different Methods of Hydration of an Alkene Draw the product formed when CH3CH2CH2CH2CH=CH2 is treated with either (a) H2O, H2SO4; or (b) BH3 followed by H2O2, HO−.arrow_forwardöaly ilaäi Give a systemic name for each compound (b) CH3CH2-0–CH(CH3)2arrow_forward
- Which of these reagent(s) will not react with HOCH2CH2CH2COOH? A) NaCN in ethanol B) C2H5OH in the presence of an acid catalyst. C) (CH3CO)2O D) Concentrated H2SO4arrow_forward(a) Draw the structure of the hemiacetal formed from one mole of benzaldehyde and one mole of ethanol. (b) Draw the structure of the acetal formed from one mole of benzaldehyde and two moles of ethanol. (c) Draw the structure of 2-methoxy-2-butanol. What compounds could you prepare this from?arrow_forwardName the following aldehydes and ketones according to IUPAC rules: lel CH3ČCH,CH,CH,ČCH,CH3 (b) CH2CH2CHO ta) CH3CH2CCHCH3 tel Id) ÇH3 H CH3CH=CHCH2CH;CH "сно CH3arrow_forward
- Draw the structure of the following compounds which parent names have been traced to a common name; (a)5-methyl-4-nitroimidazole (b)2-chloro-4-methoxythiazole.arrow_forwardc) rite in the reagent(s) over the arrow. a) C6H5N₂+ b) C6H5C=N H3C- An OH H₂C → benzene H3C benzylamine CI CH3arrow_forwardDraw structural formulas for these ketones. (a) Ethyl isopropyl ketone (b) 2-Chlorocyclohexanone (c) 2,4-Dimethyl-3-pentanone (d) Diisopropyl ketone (e) Acetone (f) 2,5-Dimethylcyclohexanonearrow_forward
- Draw the product formed when CH3CH2C=CH is treated with each of the following sets of reagents: (a) H2O, H2SO4, HgSO4; and (b) R2BH, followed by H2O2, HO−.arrow_forward1. Give the IUPAC names for the following alcohols. (a) OH OH (b) ОН (c) HO CH3CHCH2CHCHCH3 CH2CH2CCH3 -CH3 CH3 CH3 CH3 (d) (e) (f) CH3 HO HO OHarrow_forwardEthyl butyrate, CH3CH2CH2CO2CH2CH3CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring. It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l)CH3CH2CH2CO2H(l)+CH2CH3OH(l)⟶H+CH3CH2CH2CO2CH2CH3(l)+H2O(l) A chemist ran the reaction and obtained 5.40 g of ethyl butyrate. What was the percent yield, The chemist discovers a more efficient catalyst that can produce ethyl butyrate with a 78.0% yield. How many grams would be produced from 7.45g of butanoic acid and excess ethanol?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





