
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 18.5D, Problem 18.3P
Why were no products from the McLafferty rearrangement observed in the spectrum of butane-one (Figure18-3)?
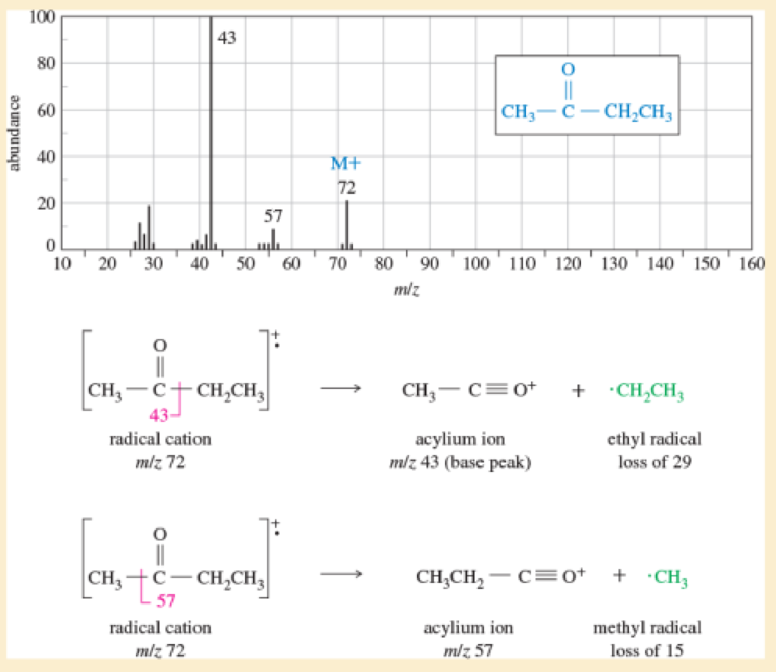
Figure 18-3
The mass spectrum of butan-2-one. Note the prominent molecular ion, together with a base peak from loss of an ethyl radical to give an acylium ion.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
the mass spectrum, IR and 13 C and 1 HNMR spectra for an unknown organic molecule. Determine the structure ofthe molecule.
How many peaks (13C) would be evident in the decoupled spectrum of
a. methylcyclohexane
b. cyclohexene
c. 1-methylcyclohexene
*show structure in the solution
4. Draw a possible molecule for each formula.
based on Formula 1
5. Which structure do you think is correct?
based on Formula 2
based on Formula 3
Chapter 18 Solutions
Organic Chemistry (9th Edition)
Ch. 18.3 - Give the IUPAC name and (if possible) a common...Ch. 18.5D - NMR spectra for two compounds are given here,...Ch. 18.5D - Why were no products from the McLafferty...Ch. 18.5D - Use equations to show the fragmentation leading to...Ch. 18.5E - Prob. 18.5PCh. 18.7D - Show how you would synthesize each compound from...Ch. 18.8 - Prob. 18.7PCh. 18.9 - Predict the products of the following reactions....Ch. 18.9 - Show how the following transformations may be...Ch. 18.10 - Prob. 18.10P
Ch. 18.11 - Show how you would accomplish the following...Ch. 18.11 - Prob. 18.12PCh. 18.12 - Propose mechanisms for a. the acid-catalyzed...Ch. 18.12 - Rank the following compounds in order of...Ch. 18.13 - Prob. 18.15PCh. 18.13 - Show how you would accomplish the following...Ch. 18.14 - Prob. 18.17PCh. 18.14 - Prob. 18.18PCh. 18.14 - Prob. 18.19PCh. 18.14 - Prob. 18.20PCh. 18.15 - 2,4-Dinitrophenylhydrazine is frequently used for...Ch. 18.15 - Prob. 18.22PCh. 18.15 - Prob. 18.23PCh. 18.16 - Prob. 18.24PCh. 18.16 - Prob. 18.25PCh. 18.16 - Show what alcohols and carbonyl compounds give the...Ch. 18.16 - In the mechanism for acetal hydrolysis shown, the...Ch. 18.16 - Prob. 18.28PCh. 18.17 - Show how you would accomplish the following...Ch. 18.18 - Prob. 18.30PCh. 18.18 - Prob. 18.31PCh. 18.18 - Prob. 18.32PCh. 18.18 - Show how Wittig reactions might be used to...Ch. 18.19 - Predict the major products of the following...Ch. 18.20C - Prob. 18.35PCh. 18.20C - Predict the major products of the following...Ch. 18 - Draw structures of the following derivatives. a....Ch. 18 - Prob. 18.38SPCh. 18 - Predict the major products of the following...Ch. 18 - Rank the following carbonyl compounds in order of...Ch. 18 - Acetals can serve as protecting groups for...Ch. 18 - Sketch the expected proton NMR spectrum of...Ch. 18 - A compound of formula C6H10O2 shows only two...Ch. 18 - The proton NMR spectrum of a compound of formula...Ch. 18 - The following compounds undergo McLafferty...Ch. 18 - An unknown compound gives a molecular ion of m/z...Ch. 18 - Show how you would accomplish the following...Ch. 18 - Prob. 18.48SPCh. 18 - Prob. 18.49SPCh. 18 - Propose mechanisms for the following reactions.Ch. 18 - Show how you would accomplish the following...Ch. 18 - Show how you would synthesize the following...Ch. 18 - Predict the products formed when cyclohexanone...Ch. 18 - Predict the products formed when...Ch. 18 - Show how you would synthesize octan-2-one from...Ch. 18 - Prob. 18.56SPCh. 18 - Both NaBH4 and NaBD4 are commercially available,...Ch. 18 - When LiAIH4 reduces 3-methylcyclopentanone, the...Ch. 18 - Prob. 18.59SPCh. 18 - Show how you would accomplish the following...Ch. 18 - There are three dioxane isomers 1,2-dioxane,...Ch. 18 - Two structures for the sugar glucose are shown on...Ch. 18 - Prob. 18.63SPCh. 18 - Prob. 18.64SPCh. 18 - Prob. 18.65SPCh. 18 - Prob. 18.66SPCh. 18 - Within each set of structures, indicate which will...Ch. 18 - Prob. 18.68SPCh. 18 - Prob. 18.69SPCh. 18 - Prob. 18.70SPCh. 18 - The UV spectrum of an unknown compound shows...Ch. 18 - a. Simple aminoacetals hydrolyze quickly and...Ch. 18 - The mass spectrum of unknown compound A shows a...Ch. 18 - Prob. 18.74SPCh. 18 - Prob. 18.75SPCh. 18 - Prob. 18.76SPCh. 18 - Prob. 18.77SP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Label the molecular ion, the base peak, and the M + 1 peak in the mass spectrum of pentane (C5H12).arrow_forwardHow many peaks can be expected in the 13C spectrum of acetylsalicylic acid (shown below)? 1 2 C THO. 7 9. +/- х 100arrow_forwardFollowing are proton decoupled 13C NMR spectra for three isomeric alcohols with the formula CAH100. A DEPT analysis yields the multiplicities shown: s=-singlet, d=doublet, t=triplet, q=quartet. Identify the alcohol responsible for each spectrum and assign each peak to an appropriate carbon atom or atoms. q A d. CDCI3 (solvent) TMS CDCI3 (solvent) TMS 190 180 170 160 150 140 130 120 110 100 90 80 70 60 50 40 30 20 10 190 180 170 160 150 140 130 120 110 100 90 80 70 60 50 40 30 20 10 O 8. B b. CDCI3 (solvent) TMS 190 180 170 160 150 140 130 120 110 100 90 80 70 60 50 40 30 20 10arrow_forward
- The two mass spectra below correspond to two isomers of C4H10O: 1-butanol and 2-butanol. Match the spectrum with the appropriate compound. Place the m/z ratio and the structures for the labeled fragments in the table below. 100 Compound 00 2 GO 40 - 1 20 - 10 20 30 40 50 60 70 80 90 100 m/ 7 100 Compound 3 80 60 40 20 0 TH 10 15 20 25 30 35 40 45 50 55 60 65 70 75 m/z Fragment 1 Fragment 2 Fragment 3 Fragment 4 m/z Fragment Relative Intensity Re-ative Intersityarrow_forward2. The mass spectrum of a hydrocarbon is shown below. Use the Rule of 13 to determine a molecular formula and use the formula to calculate units of unsaturation. Assume that the peak at m/z 120 is the molecular ion. What are possible structures for this compound? 100 80 60- 40 20 10 20 30 40 50 60 70 80 90 100 110 120 m/z Relative Intensityarrow_forward1. What do you suppose is the molecular weight of the molecule that gives the mass spectrum below? 27 Hai 55 26 45 30 40 50 Rel Abund 100r 80- 60- 40- 20- 0- m/z0 10 Rel Abund = Relative Abundance Formula 1: 20 Formula 2: Formula 3: m/z = mass divided by charge 3. What is the base peak? 2. Provide three different molecular formulas (organic molecules, please) that are possible for a molecule with the molecular weight you have chosen. What are the degrees of unsaturation (SODAR) for each? Degrees of unsaturation: Degrees of unsaturation: Degrees of unsaturation: 60 4. Draw a possible molecule for each formula. based on Formula 1 5. Which structure do you think is correct? 72 70 based on Formula 2 80 What mass is lost from the molecular ion to get to the base peak? 90 100 based on Formula 3arrow_forward
- 3. Propose a structure for the compound having the mass spectrum shown below. 100 40 20 ntt r m 80 90 100 60 70 m/z 20 30 40 50 10 110 Relative Intensityarrow_forwardB. MASS SPECTRUM Identify the following from the low resolution mass spectrum of the unknown compound: 100 Relative intensity LT. 80 80 60 40 20 Base peak 0 20 Molecular lon peak 40 Mass Spectrum of Compound P 121 152 60 C. DOUBLE BOND EQUIVALENTS Calculate the DBE of the unknown compound. m/z This information indicates, for example, the presence of 100 Mass spectrum of the compound. 120 140 160 a + xarrow_forwardWhich of the following structures corresponds to the mass spectrum shown below? Relative Intensity 100 80- 60 40 20 MS-NW-0358 0-mt 20 40 CI 60 80 100 120 m/z 140 160 180 M+ = 216 200 220arrow_forward
- In the mass spectra below, determine the Base Peak, the molecular ion peak and indicate the fragmentation process of ions marked with their respective value. 100 Mass Spectrum CH3 105 80 60 40 M 134 20 C3H100 40 80 120 160 200 240 280 m/e 100- 80 91 Mass Spectrum 60 134 40 20 149 C10 H15N 40 80 120 160 m/e 200 240 280 % of bese peak % of base peakarrow_forward12. The mass spectrum of 2,2-dimethylpropane shows only a very weak molecular ion peak at m/z = 72. However, a large peak at m/z = 57 is seen. Suggest a possible structure of the fragment giving rise to this large peak and suggest a reason as to why this peak is so large. type answerarrow_forwardIdentify the following in the given IR Mass spectrum. Compound 1 Mass Spectroscopy: Interpretation of peaks: Compound 1 Mass Spectroscopy: MASS SPECTRUM 100 80 60 40 20 0.0- 10 20 30 40 50 60 70 m/z Interpretation of peaks: Rel. Intensityarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Brooks Cole
Mass Spectrometry; Author: Professor Dave Explains;https://www.youtube.com/watch?v=hSirWciIvSg;License: Standard YouTube License, CC-BY