
Interpretation:
The
Concept Introduction:
Carboxylic acid: One
Amide: One
Amide Formation: Amide is formed when a carboxylic acid reacts with an amine or ammonia.
- Primary amide is produce when a carboxylic acid reacts with ammonia.
- Secondary and tertiary amide is produce when a carboxylic acid reacts with primary and secondary amine respectively.
- Ammonium salt is formed when tertiary amine and a carboxylic acid reacts forming an ionic compound with a carboxylate acid anion and a trialkyl ammonium cation since there is no hydrogen atom in trialkyl amine
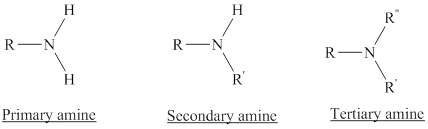
Amines are the derivatives of ammonia
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.
Aromatic Compounds: Compounds that are planar, cyclic and having
Trending nowThis is a popular solution!

Chapter 17 Solutions
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
- What is bromodeoxyuridine (BrdU) ?arrow_forwardHow can chirality and stereoisomers influence the pharmacology, bioactivity, toxicology, pharmacokinetics, and metabolism of ibuprofen? Please provide a detailed summary of what might happen if, for instance, the R enantiomer isn't able to be inverted to the bioactive S enantiomer.arrow_forwardExplain what indole alkaloids are. And describe the significance of vinflunine.arrow_forward
- What would be the characteristics of a transition-state analog for the chymotrypsin reaction?arrow_forwardDraw the structure of two different aldohexoses that yield the followingaldaric acid when oxidized with HNO3. Use Figure to name eachaldohexosearrow_forwardFrom which name reaction Lorazepam can be synthesized?arrow_forward
- List two classes of compounds derived from arachidonic acid. Suggest some reasons for the amount of biomedical research devoted to these compounds.arrow_forwardWhat makes 2, 4-dinitrophenylhydrazine suitable for characterizing aldehydes and ketones? How would carbonyl groups in the two pheromones be distinguished by a suitable chemical method?arrow_forwardCarvone exists as a pair of enantiomers. (R)-(-)-carvone smells like spearment, whereas (S)-(+)-carvone smells like caraway. Why do these enantiomers have different smells (i.e., different biological activity) ?arrow_forward
- Methadone is dispensed as its hydrochloride salt. What is the use of the salt rather than the amine?arrow_forwardGive the pharmacological property of the following alkaloids. Where do they naturally occur? morphine quinine berberine What are the chemical tests used in identifying alkaloids?arrow_forwardIn a paragraph form, provide the experimental procedures of the development of the ring structure of α-tetralene by elimination of the water molecule will result in this product being heated in the presence of strong sulphuric acid.arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





