
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 17, Problem 17.53SP
Predict the major products of the following reactions.
- a. 2,4 - dinitrochlorobenzene + NaOCH3
- b. phenol + tert - butylchloride + AlCl3
- c. nitrobenzene + fuming sulfuric acid
- d. nitrobenzene + acetylchloride + AlCl3
- e. p - methylanisole + acetylchloride + AlCl3
- f. p - methylanisole + Br2, light
- g. 1,2 - dichloro - 4 - nitrobenzene + NaNH2
- h. p - nitrotoluene +- Zn - dilute HCl
- i. p-ethylbenzenesulfonic acid + steam/H+
- j. p-ethylbenzenesulfonic acid + HNO3, H2SO4
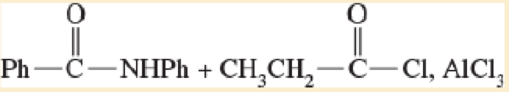
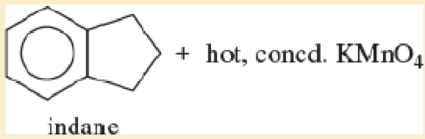
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
4. Predict the major product for the following reactions.
1. SOCI₂, pyr
2. PhMgBr
3. H
a.
b.
C.
d.
e.
f.
9.
NH₂
NH₂
OTS
CI
1. H3O*, heat
2. SOCI₂, pyr
1. HNO3, H₂SO4
2. H₂, Pt
NH₂
3. NaNO₂, HCI
4. CuCN
5. H3O*, heat
1. Br₂, FeBr3
2. SOCI₂, pyr.
1. 2
2. xs LAH
3. H₂O
1. HBr, ether
2. NaCN
3. PhMgBr
4. H*
1. NaCN
2. LiAlH4 (xs)
3. H₂O
4.
NH₂
Give the major organic product(s) of the following reactions.
A.
NO₂
NO₂
SnCl2, H3O*
B.
NO₂
NH₂
C.
NH₂
NH₂
D. B and C
E. No reaction
How would you prepare the following compounds from 2- phenylethanol? More than one step may be required.
a. Styrene (PhCH=CH2)
b. Phenylacetaldehyde (PhCH2CHO)
c. Phenylacetic acid (PhCH2CO2H)
d. Benzoic acid
e. Ethylbenzene
f. Benzaldehyde
g. 1-Phenylethanol
h. 1-Bromo-2-phenylethane
Chapter 17 Solutions
Organic Chemistry (9th Edition)
Ch. 17.1 - Prob. 17.1PCh. 17.2 - Prob. 17.2PCh. 17.3 - Prob. 17.3PCh. 17.4 - Use resonance forms to show that the dipolar sigma...Ch. 17.6A - Prob. 17.5PCh. 17.6A - Prob. 17.6PCh. 17.6B - Propose a mechanism for the brommation of...Ch. 17.6B - Prob. 17.8PCh. 17.6B - Prob. 17.9PCh. 17.7 - Prob. 17.10P
Ch. 17.8 - Draw all the resonance forms of the sigma complex...Ch. 17.9 - Predict the mononitration products of the...Ch. 17.9 - Predict the mononitration products of the...Ch. 17.9 - Prob. 17.14PCh. 17.10 - Propose products (if any) and mechanisms for the...Ch. 17.10 - Predict the products (if any) of the following...Ch. 17.10 - Which reactions will produce the desired product...Ch. 17.10 - Prob. 17.19PCh. 17.11C - Prob. 17.20PCh. 17.12A - Prob. 17.21PCh. 17.12B - Propose a mechanism that shows why p-chlorotoluene...Ch. 17.12B - Propose mechanisms and show the expected products...Ch. 17.12B - Prob. 17.24PCh. 17.13A - What products would you expect from the following...Ch. 17.13A - What organocuprate reagent would you use for the...Ch. 17.13B - What products would you expect from the following...Ch. 17.13B - Prob. 17.28PCh. 17.13C - What products would you expect from the following...Ch. 17.13C - Prob. 17.30PCh. 17.14C - Prob. 17.31PCh. 17.14C - Predict the major products of the following...Ch. 17.15A - Predict the major products of treating the...Ch. 17.15B - Prob. 17.34PCh. 17.15B - Prob. 17.35PCh. 17.15B - Predict the major products when the following...Ch. 17.15C - Prob. 17.37PCh. 17.15C - a. Based on what you know about the relative...Ch. 17.15C - Show how you would synthesize the following...Ch. 17.16A - The bombardier beetle defends itself by spraying a...Ch. 17.16B - Predict the products formed when m-cresol...Ch. 17.16B - Prob. 17.42PCh. 17.16B - Prob. 17.43PCh. 17.16B - Predict the site(s) of electophilic attack on...Ch. 17.16B - Prob. 17.45PCh. 17.16B - Prob. 17.46PCh. 17.16B - Propose a synthetic sequence of this...Ch. 17.16B - Prob. 17.48PCh. 17.16B - Starting from toluene, propose a synthesis of this...Ch. 17 - Prob. 17.50SPCh. 17 - Prob. 17.51SPCh. 17 - Show how you would synthesize the following...Ch. 17 - Predict the major products of the following...Ch. 17 - Predict the major products of bromination of the...Ch. 17 - What products would you expect from the following...Ch. 17 - Prob. 17.56SPCh. 17 - Prob. 17.57SPCh. 17 - The following compound reacts with a hot,...Ch. 17 - Prob. 17.59SPCh. 17 - Electrophilic aromatic substitution usually occurs...Ch. 17 - Prob. 17.62SPCh. 17 - The most common selective herbicide for killing...Ch. 17 - Furan undergoes electrophilic aromatic...Ch. 17 - Prob. 17.65SPCh. 17 - Bisphenol A is an important component of many...Ch. 17 - Prob. 17.67SPCh. 17 - Prob. 17.68SPCh. 17 - Prob. 17.69SPCh. 17 - In Chapter14, we saw that Agent Orange contains...Ch. 17 - Phenol reacts with three equivalents of bromine in...Ch. 17 - Prob. 17.72SPCh. 17 - Prob. 17.73SPCh. 17 - A common illicit synthesis of methamphetamine...Ch. 17 - Prob. 17.75SPCh. 17 - Prob. 17.76SPCh. 17 - Prob. 17.77SPCh. 17 - Prob. 17.78SP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Which of the provided schemes can be used to synthesize p-chlorophenol p-chlorophenol from benzen e? A. None of the below schemes are correct B. O C. D. SO3 H₂SO4 Cl₂ FeCl 903 H₂SO4 Product Product Product 1. NaOH 2. H₂O* SO 3 H₂SO4 Cl₂ FeCl Product Product Product Cl₂ FeCl H₂/Pd 1. NaOH 2. H₂O* p-Chlorophenol p-Chlorophenol p-Chlorophenolarrow_forwardFrom the table of reagents shown below, show how you synthesize the product from the given reactant. OCH₂CH3 OH a. NBS, (PhCO₂)2 b. Br2, FeBr3 c. Br₂, H+ d. OH e. NaOH(s), A f. HNO3, H₂SO4 g. H₂ CrO4 Reagents available h. CH3 CI, AICI, o. CuCN i. CH3 CH₂ Br p. H₂O+, A j. CH₂O, A q. CH3 CH₂ COCI, AICI, k. CH3COCI, AICI3 r. H₂NNH₂, OH, A 1. Mg, Et₂ O s. H₂, Pd/C m. CO₂ (s) then H3O+ t. H₂PO₂ n. HONO 0°C u. Cl₂, FeCl3 (Enter the letter(s) of the reagent(s) needed in the box, in the order that they must be used. No more than two steps are required to perform the synthesis. Note that a retrosynthetic arrow is used, and therefore the reactant is shown on the right.) Answer:arrow_forward6. Provide the structure for the major product in the following reactions. b. P f. OH g. Lia * Oia ملی CI 1. SOCI₂, pyr. 2. to Br 2 1. LIAIH4 (xs) 2. H₂O 1. LIAI(OR) 3H 2. H₂O 2. NH₂ Et₂CuLi 1. EtMgBr (xs) 2. H₂O 1. Mg 2. CO₂ 3. H* 4. SOCI₂, pyr 1. [H], NaBH3CN, EtNH₂ CI pyridinearrow_forward
- What product is obtained from the reaction of excess benzene with each of the following reagents? a. isobutyl chloride + AlCl3 b. 1-chloro-2,2-dimethylpropane + AlCl3 c. dichloromethane + AlCl3arrow_forwardFrom the table of reagents shown below, show how you synthesize the product from the given reactant. Br Reagents available a. NBS, (PhCO2)2 h. CH3 Cl, AIC13 CUCN 0. b. Br2, FeBr3 i. CH3 CH2 Br р. НзО*, д с. Вг2, Н+ j. CH3O¯, A q. CH; CH2 COCI, AICI3 r. k. CH3 COCI, AICI3 H2NNH2, OH, A d. OH- e. NaOH(s), A 1. Mg, Et20 s. H2, Pd/C f. HNO3, H,SO4 m. CO2(s) then H3O+ t. H3PO2 g. H2 CrO4 n. HONO 0°C u. Cl2, FeCl3 (Enter the letter(s) of the reagent(s) needed in the box, in the order that they must be used. No more than two steps are required to perform the synthesis. Note that a retrosynthetic arrow is used, and therefore the reactant is shown on the right.) Answer:arrow_forwardPredict the major products of each of the following reactions. CH3 Br,/CCI4 HO a. e. CH3 HNO3 Br2 f. OMe Br,/CCI4 С. Br2 g. NO2 OMe H2SO4 d. high Temp OMe b.arrow_forward
- 2. Draw products for the following reactions and provide the names for each reaction shown. Include dashes and wedges when needed A. B. C. D. E. Reaction H₂ Pt H₂O H+ HBr 2- Br₂ CC14 Br₂ H₂O Namearrow_forward12. Which reagents/conditions would lead to the transformation depicted below in high yield? a. NaOMe, MeOH b. MeOH, H₂O* c. NaOEt, EtOH 13. In the structure of 2-fluoropropanoic acid shown, the oxidation number on C1 is the oxidation number on C2 is a. +1, +2, -3 b. -3, +1, +3 d. NaOH, H₂O e. two of these , and the oxidation number on C3 is, c. 0, 0, 0 d. +3,0,-3 e. +2,0, -2 ??? OH 14. What will be the outcome of the following reaction sequence? B. 1. NaOEt, EtOH 2. PCC 3. isopropylMgBr 4. H₂0¹ ??? a. 2-ethyl-3-isopropyl-2,3-dimethyloxirane b. 2,3,4-trimethyl-3,4-epoxyhexane c. 1,2,3,4-tetramethyl-2,3-epoxypentane d. Both a and b are suitable. e. All of the above are correct. OH 15. What is a suitable IUPAC name for the structure shown at the right? OH D. F H OH LOH CH3 3 OHarrow_forwardPredict the major product for the following reaction. A. B. ze ze 1. (Et)₂CuLi 2. H3O+ C. D. ze OHarrow_forward
- Which starting material produces the two products shown after ozonolysis and a reductive workup? 1. O3 2. DMS C. А. D. В. A.arrow_forward6. Predict the product for the following reaction. "NH₂ Br H₂N NO₂ D. H₂O* A. Fe/HCI IV OH OH Br 11 NH₂ "NH₂ H₂N A. IB. II C. III D. IV E. V 7. Provide the reagents necessary to carry out the following reaction. NH₂ V 1. LIAIH4 2. H₂O NH₂ B. H₂CCOCI C. Br₂/FeBr3 E. dil. Na₂CO3 IIIarrow_forwardFrom the table of reagents shown below, show how you synthesize the product from the given reactant. H3C NO₂ a. NBS, (PhCO₂), b. Bra, FeBr, c. Br₂, H d. OH e. NaOH(s), A 1. HNO₂, H₂SO 9. H₂ CrO Answer: NO₂ Reagents available. h. CH, CI, AICI, o. CuCN 1. CH₂ CH₂ Br p. H₂O, A 1. CH₂O, A q. CH₂ CH₂ COCI, AICI, k. CH, COCI, AICI r. H₂NNH,, OH A 1. Mg, Et₂0 s. H₂. Pd/C m. CO₂ (s) then H₂0 t. H₂PO, n. HONO 0°C u. Cl₂, FeCla (Enter the letter(s) of the reagent(s) needed in the box, in the order that they must be used. No more than two steps are required to perform the synthesis. Note that a retrosynthetic arrow is used, and therefore the reactant is shown on the right.) Previous Nextarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
IR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=_TmevMf-Zgs;License: Standard YouTube License, CC-BY