
Concept explainers
(a)
Interpretation:
The type of carbonyl group in erythrulose should be identified.
Concept introduction:
An aldehyde has an oxygen atom double bonded to carbon and a hydrogen atom bonded to the same carbon.
A ketone has oxygen atom double bonded to the C and there are no H atoms bonded to it.
In both aldehydes and ketones this carbon is known as carbonyl carbon.
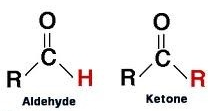
R − Other atoms or atomic groups connected to the compound apart from the carbonyl group.
(b)
Interpretation:
The 3 hydroxyl groups should be classified as primary (1
Concept introduction:
Depending on the number of hydrogen atoms attached to the carbon atom with hydroxyl group (-OH), they can be classified as primary (1
(c)
Interpretation:
Out of the 2 functional groups in erythrulose, which functional group is reacting with Tollens reagent should be considered.
Concept introduction:
Tollens reagent is amixture of a silver nitrate solution, ammonia and some sodium hydroxide. NaOH is added to maintain the pH in a basic level as the Tollensreaction only happens in a basic medium.
(d)
Interpretation:
Out of the 2 functional groups in erythrulose, which functional group is reacting with K2Cr2O7 should be considered.
Concept introduction:
K2Cr2O7 is an oxidizing agent. It has the ability to oxidize some functional groups of an organic compound.
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
General, Organic, and Biological Chemistry - 4th edition
- Prednisolone is a synthetic corticosteroid that is used as an anti-inflammatory agent. Sudikel НО НО HO Hi H. B Which box contains hydroxyl group? A. Воx A Вох В botharrow_forwardD-Arabinose can exist in both pyranose and furanose forms.a. Draw the a and ß anomers of D-arabinofuranose.b. Draw the a and ß anomers of D-arabinopyranosearrow_forwardstructure of each type of compound. A. D –aldotriose B. L-ketohexosearrow_forward
- Which structure of Erythrose is referred to by the given Fischer projection? A. L-Erythrose B. D-Erythrose но- -H HO H CH2OHarrow_forwarda. How many stereogenic centers are present in α-D-galactose? b. Label the hemiacetal carbon in α-D-galactose. c. Draw the structure of β-D-galactose. d. Draw the structure of the polyhydroxy aldehyde that cyclizes to α- and β-D-galactose.arrow_forwardExplain Tollens reagent ?arrow_forward
- Please draw the structure Aarrow_forward3. A(n) is a molecule that is synthesized in one part of the body and has an effect in a different part. A) fatty acid B) enzyme C) hormone D) vitamin 4. DNA is a double-stranded nucleic acid that exists as a double helix. Which is responsible for holding the double helix of DNA together? A) Phosphodiester bonds between complementary base pairs on each strand B) B-N-glycosidic linkages between the sugar-phosphate backbone of each strand C) Disulfide bonds between the cysteine residues on each strand D) Hydrogen bonding between complementary bases on each strand 5. Which type of inhibitor is most likely to bind somewhere other than an enzyme's active site? A) competitive B) lock and key C) noncompetitive D) substrate 6. The compound shown below is best classified as what type of lipid? HO, A) Triacylglycerol B) Soap C) Fatty acid D) Waxarrow_forwardWhich of the following area. hemiacetals? b. acetals? c. hydrates?arrow_forward
- a. Reaction of acyl compounds with LiAlH4 forms alcohols. b.Reaction of acyl compounds with alcohols forms esters. c.Reaction of carbonyl compounds with LiAlH4 forms alcohols. d.Reaction of carbonyl compounds with alcohols forms hemiacetals. Which are false?arrow_forwardQuestion 2: Draw the organic products formed in each hydrogenation. 1arrow_forwardFructose, present with glucose in honey, reacts with Benedicts reagent. Circle the structural features that enable fructose to react.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning



