
(a)
Interpretation:
The laboratory synthesis of
Concept introduction:
The replacement of hydrogen atom attached to a carbon atom of electron-rich benzene ring by an incoming electrophile is known as electrophilic
Answer to Problem 16.43AP
The laboratory synthesis of
Explanation of Solution
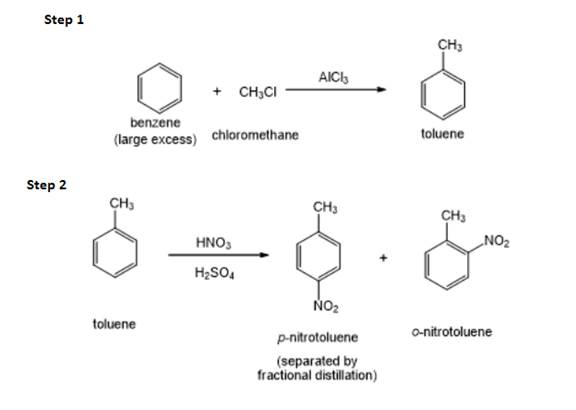
The structure of
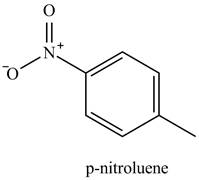
Figure 1
The methyl is an ortho and para directing group and nitro is a meta directing group. the compound is para compound. Therefore, the benzene will first undergo methylation reaction with chloromethane and
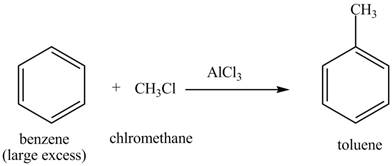
Figure 2
The toluene will undergo nitration reaction with nitric acid in sulfuric acid to from ortho and para-substituted compounds. The para-substituted gets separated from ortho compound with the help of fractional distillation process. The corresponding chemical reaction is shown below.
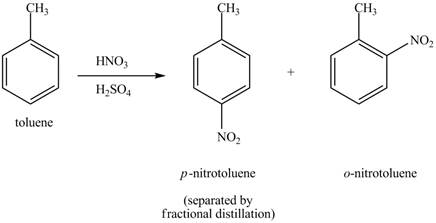
Figure 3
The laboratory synthesis of
(b)
Interpretation:
The laboratory synthesis of
Concept introduction:
The replacement of hydrogen atom attached to a carbon atom of electron-rich benzene ring by an incoming electrophile is known as electrophilic aromatic substitution reaction. The rate of electrophilic aromatic substitution reaction depends on the substituted group on the aromatic ring. The ring deactivating group retards the electrophilic aromatic substitution reaction and ring activating group enhances the electrophilic aromatic substitution reaction.
Answer to Problem 16.43AP
The laboratory synthesis of
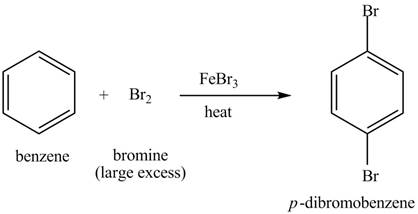
Explanation of Solution
The structure of
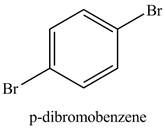
Figure 4
Benzene reacts with an excess of bromine gas in the presence of a
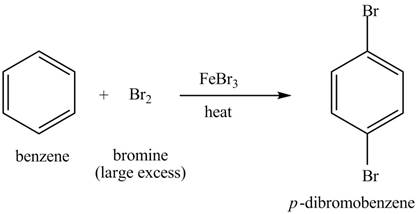
Figure 5
The laboratory synthesis of
(c)
Interpretation:
The laboratory synthesis of
Concept introduction:
The replacement of hydrogen atom attached to a carbon atom of electron-rich benzene ring by an incoming electrophile is known as electrophilic aromatic substitution reaction. The rate of electrophilic aromatic substitution reaction depends on the substituted group on the aromatic ring. The ring deactivating group retards the electrophilic aromatic substitution reaction and ring activating group enhances the electrophilic aromatic substitution reaction.
Answer to Problem 16.43AP
The laboratory synthesis of
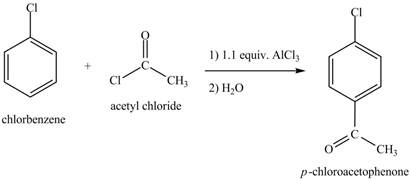
Explanation of Solution
The structure of
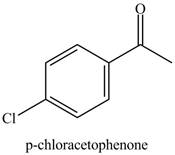
Figure 6
Benzene reacts with chlorine gas in the presence of a catalyst
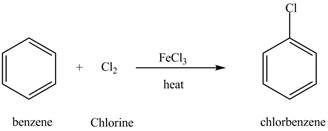
Figure 7
The chlorobenzene undergoes Friedel Craft acylation reaction with acetyl chloride in the presence of
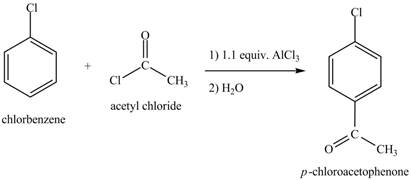
Figure 8
The laboratory synthesis of
(d)
Interpretation:
The laboratory synthesis of
Concept introduction:
The replacement of hydrogen atom attached to a carbon atom of electron-rich benzene ring by an incoming electrophile is known as electrophilic aromatic substitution reaction. The rate of electrophilic aromatic substitution reaction depends on the substituted group on the aromatic ring. The ring deactivating group retards the electrophilic aromatic substitution reaction and ring activating group enhances the electrophilic aromatic substitution reaction.
Answer to Problem 16.43AP
The laboratory synthesis of
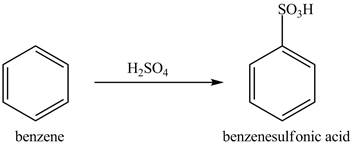
Explanation of Solution
The structure of
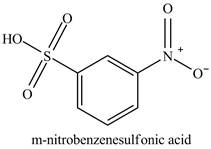
Figure 9
The benzene molecule will undergo sulfonation reaction with sulfuric acid. The electrophile
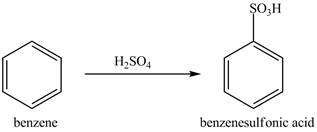
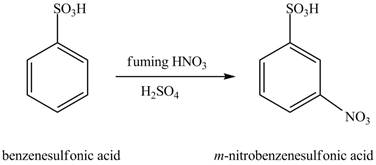
Figure 10
The benzenesulfonic acid will undergo nitration reaction with fuming nitric acid in sulfuric acid to form
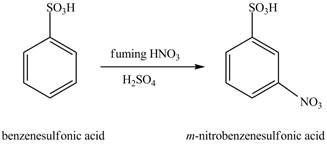
Figure 11
The laboratory synthesis of
(e)
Interpretation:
The laboratory synthesis of
Concept introduction:
The replacement of hydrogen atom attached to a carbon atom of electron-rich benzene ring by an incoming electrophile is known as electrophilic aromatic substitution reaction. The rate of electrophilic aromatic substitution reaction depends on the substituted group on the aromatic ring. The ring deactivating group retards the electrophilic aromatic substitution reaction and ring activating group enhances the electrophilic aromatic substitution reaction.
Answer to Problem 16.43AP
The laboratory synthesis of
Explanation of Solution
The structure of
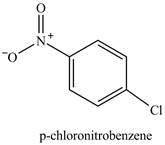
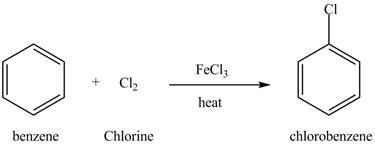
Figure 12
Benzene reacts with chlorine gas in the presence of a catalyst
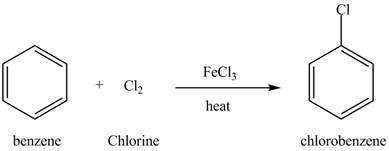
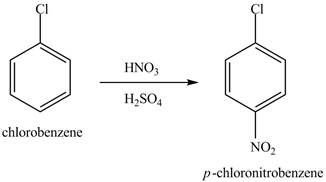
Figure 13
The chlorobenzene will undergo nitration reaction with nitric acid in sulfuric acid to form
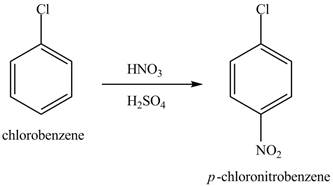
Figure 14
The laboratory synthesis of
(f)
Interpretation:
The laboratory synthesis of
Concept introduction:
The replacement of hydrogen atom attached to a carbon atom of electron-rich benzene ring by an incoming electrophile is known as electrophilic aromatic substitution reaction. The rate of electrophilic aromatic substitution reaction depends on the substituted group on the aromatic ring. The ring deactivating group retards the electrophilic aromatic substitution reaction and ring activating group enhances the electrophilic aromatic substitution reaction.
Answer to Problem 16.43AP
The laboratory synthesis of
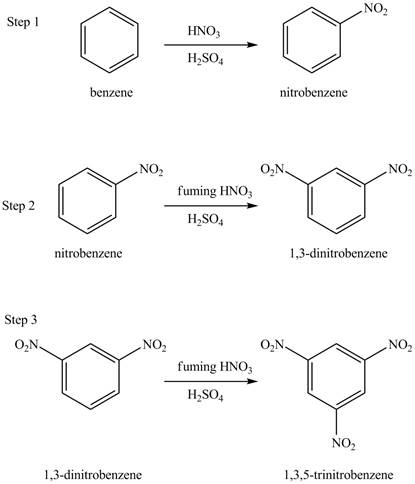
Explanation of Solution
The structure of
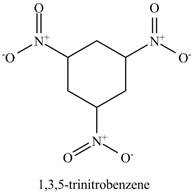
Figure 15
The benzene will undergo nitration reaction with nitric acid in sulfuric acid to form nitrobenzene. The nitro group is a ring deactivating group and meta directing group. Therefore, some strong condition is required to substitute another electrophile on it. The nitrobenzene reacts with fuming nitric acid and sulfuric acid to form
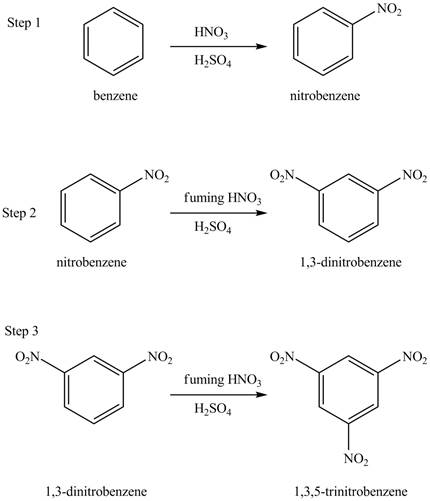
Figure 16
The laboratory synthesis of
(g)
Interpretation:
The laboratory synthesis of
Concept introduction:
The replacement of hydrogen atom attached to a carbon atom of electron-rich benzene ring by an incoming electrophile is known as electrophilic aromatic substitution reaction. The rate of electrophilic aromatic substitution reaction depends on the substituted group on the aromatic ring. The ring deactivating group retards the electrophilic aromatic substitution reaction and ring activating group enhances the electrophilic aromatic substitution reaction.
Answer to Problem 16.43AP
The laboratory synthesis of
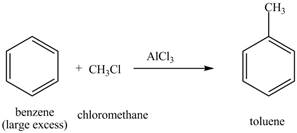
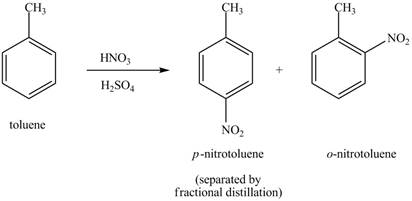
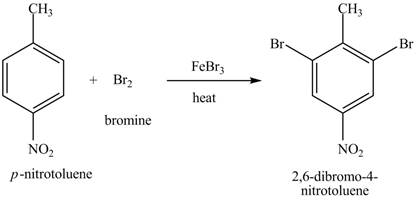
Explanation of Solution
The structure of
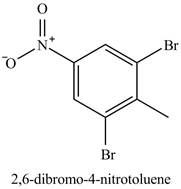
Figure 17
The benzene will first undergo methylation reaction with chloromethane and
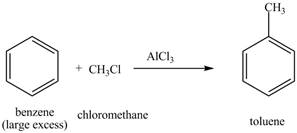
Figure 18
The toluene will undergo nitration reaction with nitric acid in sulfuric acid to form ortho and para-substituted compounds. The para-substituted gets separated from ortho compound with the help of fractional distillation process. The corresponding chemical reaction is shown below.
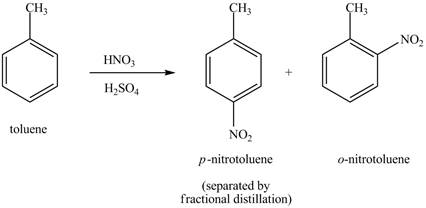
Figure 19
The compound
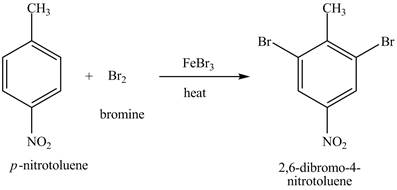
Figure 20
The laboratory synthesis of
(h)
Interpretation:
The laboratory synthesis of
Concept introduction:
The replacement of hydrogen atom attached to a carbon atom of electron-rich benzene ring by an incoming electrophile is known as electrophilic aromatic substitution reaction. The rate of electrophilic aromatic substitution reaction depends on the substituted group on the aromatic ring. The ring deactivating group retards the electrophilic aromatic substitution reaction and ring activating group enhances the electrophilic aromatic substitution reaction.
Answer to Problem 16.43AP
The laboratory synthesis of
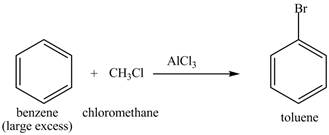
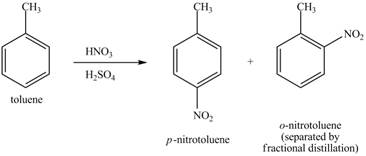
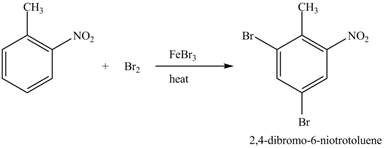
Explanation of Solution
The structure of
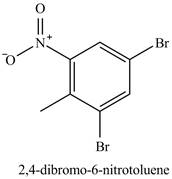
Figure 21
The benzene will first undergo methylation reaction with chloromethane and
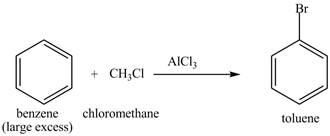
Figure 22
The toluene will undergo nitration reaction with nitric acid in sulfuric acid to form ortho and para-substituted compounds. The para-substituted gets separated from ortho compound with the help of fractional distillation process. The corresponding chemical reaction is shown below.

Figure 23
The compound
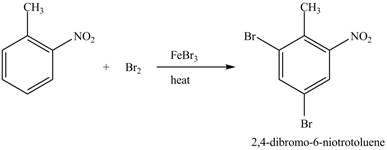
Figure 24
The laboratory synthesis of
(i)
Interpretation:
The laboratory synthesis of
Concept introduction:
The replacement of hydrogen atom attached to a carbon atom of electron-rich benzene ring by an incoming electrophile is known as electrophilic aromatic substitution reaction. The rate of electrophilic aromatic substitution reaction depends on the substituted group on the aromatic ring. The ring deactivating group retards the electrophilic aromatic substitution reaction and ring activating group enhances the electrophilic aromatic substitution reaction.
Answer to Problem 16.43AP
The laboratory synthesis of
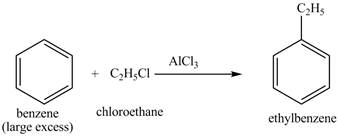
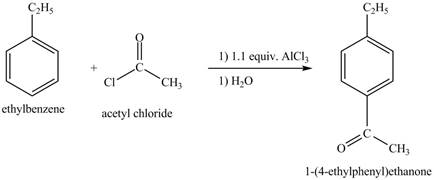
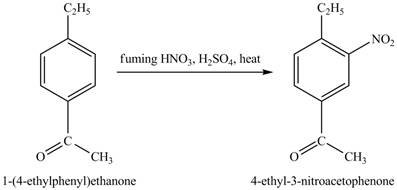
Explanation of Solution
The structure of
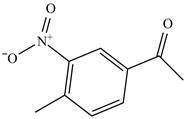
Figure 25
The benzene will first undergo ethylation reaction with chloromethane and
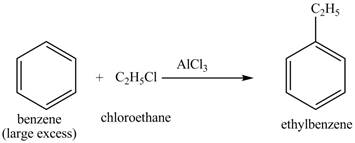
Figure 26
The ethylbenzene undergoes Friedel Craft acylation reaction with acetyl chloride in the presence of
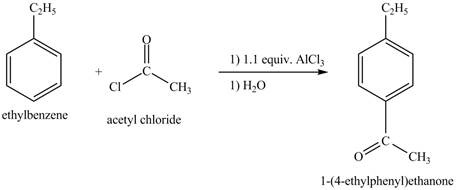
Figure 27
The compound
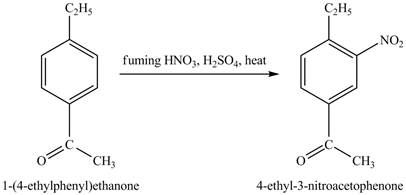
Figure 28
The laboratory synthesis of
(j)
Interpretation:
The laboratory synthesis of cyclopentylbenzene from benzene and any other reagents is to be predicted.
Concept introduction:
The replacement of hydrogen atom attached to a carbon atom of electron-rich benzene ring by an incoming electrophile is known as electrophilic aromatic substitution reaction. The rate of electrophilic aromatic substitution reaction depends on the substituted group on the aromatic ring. The ring deactivating group retards the electrophilic aromatic substitution reaction and ring activating group enhances the electrophilic aromatic substitution reaction.
Answer to Problem 16.43AP
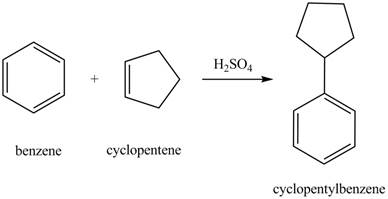
The laboratory synthesis of cyclopentylbenzene from benzene and any other reagents is shown below.
Explanation of Solution
The structure of cyclopentylbenzene is shown below.
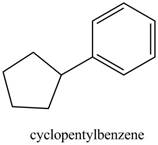
Figure 29
Benzene reacts with cyclopentene in the presence of sulfuric acid to form cyclopentyl benzene. The sulfuric acid acts as a catalyst to generate carbocation from cyclopentene. This carbonation acts as an electrophile and attacks the benzene ring. The corresponding chemical reaction is shown below.

Figure 30
The laboratory synthesis of cyclopentylbenzene from benzene and any other reagents is shown in Figure 30.
Want to see more full solutions like this?
Chapter 16 Solutions
Organic Chemistry Study Guide and Solutions
- Explain the following statements. You must use chemical equations to justify your explanation. (ii) (I) Phenol is more acidic than cyclohexanol.arrow_forwardOutline syntheses of each of the following from aniline and any necessary organic or inorganic reagents. (a) p-Nitroaniline (b) 2,4-Dinitroaniline (c) p-Aminoacetanilidearrow_forward18.18 Provide a structural formula for the product from each of the following reactions. (a) (c) (e (1) LDA (2) CH,CH,I NaH CI (b) (d) O. (f Br, CH,CO,H Br, (excess), NaOHarrow_forward
- (b) Distinguish each as aromatic or antiaromatic in terms of electronic basis. Explain which one is more stable. (i) Compound A Compound B (ii) Compound C Compound D (iii) Compound E Compound Farrow_forward(a) Which is more reactive towards Electrophilic aromatic substitution (EAS)? (b) Which of the following is a a meta-substituted compound? (c) Which is the least reactive towards Electrophilic aromatic substitution (EAS)? (d) Which is an ortho-substituted compound?arrow_forwardWhich is the stronger acid in each of the following pairs? Explain your reasoning. (a) Phenol or p-hydroxybenzaldehyde (b) m-Cyanophenol or p-cyanophenol (c) o-Fluorophenol or p-fluorophenolarrow_forward
- 3) Draw equations of the following reactions and and explain to which direction is the respective quillibrium shifted. a) cyclohexylamine + water b) aniline + sulphuric acid c) triethylamine + acetic acidarrow_forwardAnswer ALL parts of this question. (a) Using resonance structures, discuss the following statement; phenols are much stronger acids than aliphatic alcohols. (b) Give the structure of a stronger organic acid than phenol. (c) Rationalise the acidity of the latter organic acid by drawing two resonance structures of the conjugate base. (d) Eugenol is a natural product derived from the dried flower buds of the evergreen tree, Eugenia aromatica. Briefly describe a procedure with reagents required to extract eugenol as a single component from the mixture of compounds present in these flower buds. OH LOCH 3 eugenolarrow_forward(b) Suggest a reasonable biosynthesis for the naturally occurring alkaloid coniine (isolated from hemlock), starting from octanoic acid. Coniinearrow_forward
- (a) (From benzene)arrow_forwardProvide the reagents and solvents (where appropriate) needed to bring about the following transformations. (a) CI (b)arrow_forwardShow how you can synthesize the following compounds starting with benzene, toluene, and alcohols containing no morethan four carbon atoms as your organic starting materials. Assume that para is the major product (and separable fromortho) in ortho, para mixtures.(a) pentan-1-amine (b) N-methylbutan-1-aminearrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





