
Concept explainers
Interpretation:
The structural formula for the compound D and for the product,
Concept introduction:
The
Wittig reaction is a reaction in which a nucleophilic elimination occurs after nucleophilic addition reaction. In this reaction an aldehyde or ketone reacts with a triphenyl phosphonium ylide (Wittig reagent) to give an
The reaction can be represented as shown below,
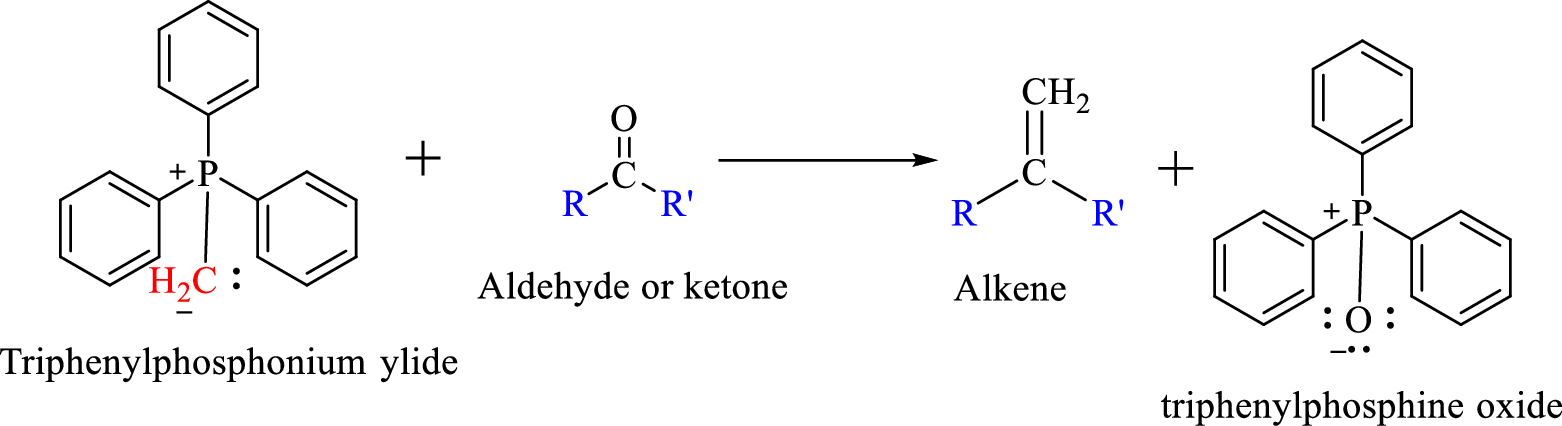
Mechanism:
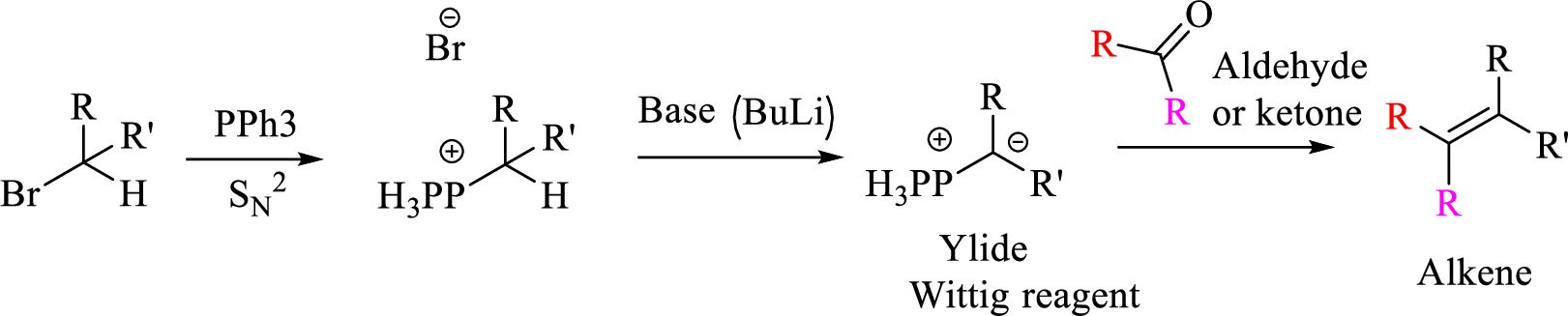
It is possible to generate sulphur ylides in a manner similar to that used to produce phosphonium ylides. For example, treating a sulfonium salt with a strong base gives the sulphur ylide.
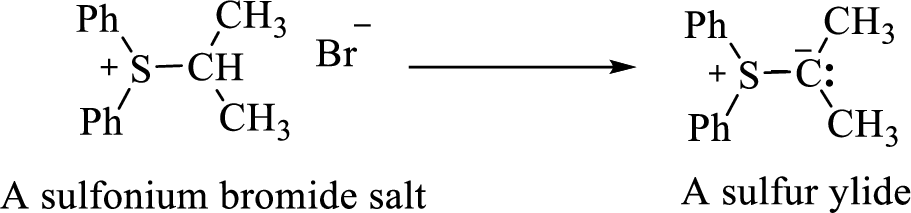
Sulfur ylides reacts with ketone to give
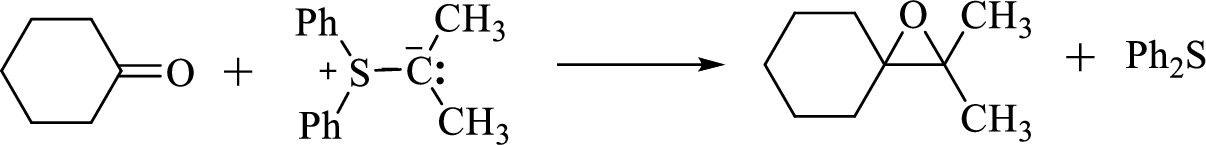
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
Organic Chemistry
- Following is the structural formula of the tranquilizer meparfynol (Oblivon). Propose a synthesis for this compound starting with acetylene and a ketone. (Notice the -yn- and -ol in the chemical name of this compound, indicating that it contains alkyne and hydroxyl functional groups.)arrow_forwardNonconjugated , -unsaturated ketones, such as 3-cyclohexenone, are in an acid-catalyzed equilibrium with their conjugated , -unsaturated isomers. Propose a mechanism for this isomerization.arrow_forwardGrignard reagent is a versatile tool in synthetic organic chemistry. Using bromocyclopentane as a starting material, show how a Grignard reagent, X, is synthesized. Reaction of X with water produces compound Y while treatment in carbon dioxide followed by hydrolysis forms compound Z. 3-methyl-2butanone reacts with X and hydrolyses to yield compound AA. Draw the structural formulae of compounds Y, Z and AA and write the chemical equations respectively.arrow_forward
- Provide reagents/conditions to accomplish the following syntheses. More than one step is required in some cases.arrow_forwardChemistry Provide reagents/conditions to accomplish the following syntheses. Several steps are required in some cases.arrow_forwardPentane-2,4- dione reacts with NaOH to give H2O and sodium salt of a carbaion. write a complete structural formula for the carbanion and use resonance forms to show stabilization of the carbanion.arrow_forward
- Following are 1H-NMR spectra for compounds B (C6H12O2) and C (C6H10O). Upon warming in dilute acid, compound B is converted to compound C. Deduce the structural formulas for compounds B and C.arrow_forwardHydroboration-oxidation of 2-pentyne gives a mixture of two ketones, each with the molecular formula C5H10O. Propose structural formulas for these two ketones and for the enol from which each is derived.arrow_forwardWhen A is reacted with hot aqueous NaOH, a compound B of molecular formula C8H11NO is produced. With this information, write the correct structure of B and propose the reaction mechanism (step by step, with the correct use of arrows) to obtain B.arrow_forward
- Syntheses of each of the following compounds have been reported in the chemical literature. Using the indicated starting material and any necessary organic or inorganic reagents, describe short sequences of reactions that would be appropriate for each transformation. (a) 1,1,5-Trimethylcyclononane from 5,5-dimethylcyclononanonearrow_forward3) Optically active alkyl bromide A (CÂH₁7Br) reactions with potassium tert-butoxide in tert- butanol to yield compound B (CH₁6). Compound B affords the carbonyl compound shown below and formaldehyde when subjected to ozonolysis followed by a reductive workup. Write the reactions and draw the structures of A & B. شدarrow_forwardPlease provide the reagents for the following transformations.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

