
Concept explainers
(a)
Interpretation:
The product obtained on reaction of
Concept introduction:
Conjugated dienes are defined as two double bonds separated by one single bond. They are more stable than non-conjugated double bonds due to resonance and hybridization energy. Along with that the heat of hydrogenation effects the stability of conjugated dienes. It states that higher the heat of hydrogenation energy lowers the stability of dienes.
Answer to Problem 15.43AP
The complete reaction is written as shown below.
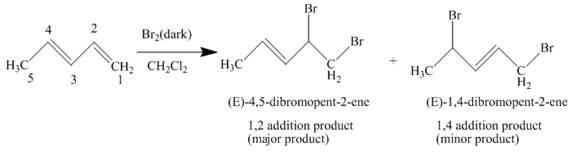
Explanation of Solution
When
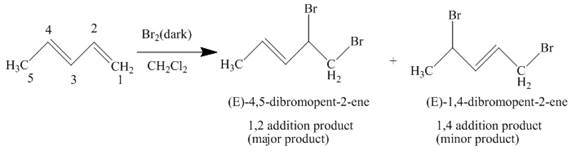
Figure 1
The product obtained on reaction of
(b)
Interpretation:
The product obtained on reaction of
Concept introduction:
Conjugated dienes areb defined as two double bonds separated by one single bond. They are more stable than non-conjugated double bonds due to resonance and hybridization energy. Along with that the heat of hydrogenation effects the stability of conjugated dienes. It states that higher the heat of hydrogenation energy lowers the stability of dienes.
Answer to Problem 15.43AP
The complete reaction is written as shown below.
Explanation of Solution
When

Figure 2
The product obtained on reaction of
(c)
Interpretation:
The product obtained on reaction of
Concept introduction:
Conjugated dienes are defined as two double bonds separated by one single bond. They are more stable than non-conjugated double bonds due to resonance and hybridization energy. Along with that the heat of hydrogenation effects the stability of conjugated dienes. It states that higher the heat of hydrogenation energy lowers the stability of dienes.
Answer to Problem 15.43AP
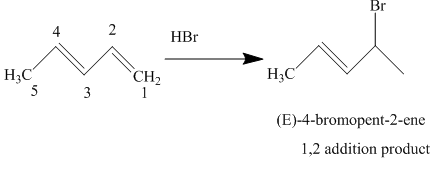
The complete reaction is written as shown below.
Explanation of Solution
When

Figure 3
The product obtained on reaction of
(d)
Interpretation:
The product obtained on reaction of
Concept introduction:
Conjugated dienes are defined as two double bonds separated by one single bond. They are more stable than non-conjugated double bonds due to resonance and hybridization energy. Along with that the heat of hydrogenation effects the stability of conjugated dienes. It states that higher the heat of hydrogenation energy lowers the stability of dienes.
Answer to Problem 15.43AP
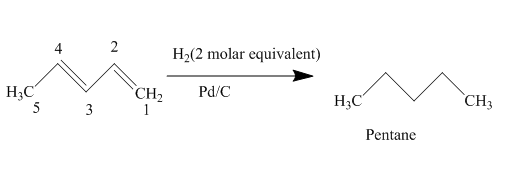
The complete reaction is written as shown below.
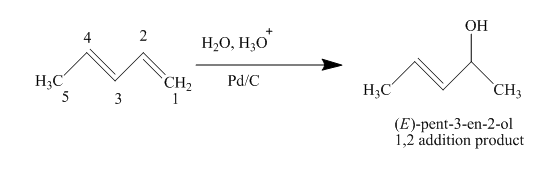
Explanation of Solution
When

Figure 4
The product obtained on reaction of
(e)
Interpretation:
The product obtained on reaction of
Concept introduction:
Conjugated dienes are defined as two double bonds separated by one single bond. They are more stable than non-conjugated double bonds due to resonance and hybridization energy. Along with that the heat of hydrogenation effects the stability of conjugated dienes. It states that higher the heat of hydrogenation energy lowers the stability of dienes.
Answer to Problem 15.43AP
The reaction does not undergo any chemical change because
Explanation of Solution
When
Alkenes do not undergo any nucleophilic addition reaction it means no product is formed in this reaction.
(f)
Interpretation:
The product obtained on reaction of
Concept introduction:
A chemical reaction that involves cycloaddition is known as Diels-Alder reaction. The reactant molecules that give rise to the product are diene and dienophile. The
Answer to Problem 15.43AP
The complete reaction is written below.
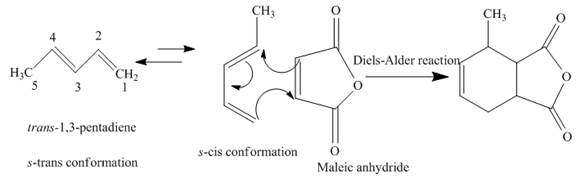
Explanation of Solution
When

Figure 5
The product obtained on reaction of
Want to see more full solutions like this?
Chapter 15 Solutions
Organic Chemistry Study Guide and Solutions
- Provide the major product for the following reaction? (1) BH3, ether (2) H2O2, OHarrow_forward(a) (b) HO N N-H Catalytic H+ (-H₂O) ? (c) NH2 HO N (d)arrow_forwardNeopentyl alcohol, (CH3)3CCH2OH, reacts with concentrated HBr to give 2-bromo2-methylbutane, a rearranged product. Propose a mechanism for the formation of thisproduct.arrow_forward
- Which of the following compounds are suitable solvents for Grignard reactions?(a) n-hexane (b) CH3¬O¬CH3 (c) CHCl3(d) cyclohexane (e) benzene (f) CH3OCH2CH2OCH3arrow_forwardWhat is the expected product for the following reaction? А (a) (c) OH OH hot KMnO4 H,0+ (b) НО (d) ? ОН OHarrow_forward1. How would you prepare the following alkyl halides from the appropriate alcohols? (a) ÇI (b) Br CH3 CH3CH3 CH;CHCH,CHCH3 ČH3 2. Predict the products of the following reaction: (a) он CH3 (b) H2C CH;CH,CHCH,CHCH, Per, socl2. PBr3 ? H3Carrow_forward
- When cis-4-chlorocyclohexanol is treated with sodium hydroxide in ethanol, it gives mainly the substitution product trans-1,4-cyclohexanediol (1). Under the same reaction conditions, trans-4-chlorocyclohexanol gives 3-cyclohexenol (2) and the bicyclic ether (3). (a) Propose a mechanism for formation of product (1), and account for its configuration. (b) Propose a mechanism for formation of product (2). (c) Account for the fact that the bicyclic ether (3) is formed from the trans isomer but not from the cis isomer.arrow_forwardOne frequently used method for preparing methyl esters is by reaction of carboxylic acids with diazomethane, CH2N2. The reaction occurs in two steps: (l) protonation of diazomethane by the carboxylic acid to yield methyldiazonium ion, CH3N2+, plus a carboxylate ion; and (2) reaction of the carboxylate ion with CH3N2+. (a) Draw two resonance structures of diazomethane, and account for step 1. (b) What kind of reaction occurs in step 2?arrow_forward(b) Predict the suitable solvent (H2O or CH3COCH3) to increase the reaction of bromopropane (CH3CH2CH2B1) with sodium hydroxide (NaOH). Two reactions are shown below: NaOH, 55 °C CH;CH,CH,Br CH;CH,CH,OH + NaBr H,O (i) NaOH, 55 °C CH;CH,CH,Br CH;CH,CH,OH NaBr H,C CH (ii)arrow_forward
- 8a)Provide mechanisms for the following reactions:arrow_forwardWhich set of reagents would effect the conversion, OH (а) ВH3:THF, then H2O2/HO- (b) Hg(OAc)2, THF-H2O, then NaBH4/HO¯ (c) H3O+, H2O (d) More than one of these (e) None of thesearrow_forward(b) Complete the following reactions to show the product(s) formed: (i) NaCN + (ii) 2 NH3 e =O R R -CI OH DMSO Brarrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

