
Concept explainers
(a)
Interpretation:
To indicate whether B vitamin niacin is involved as a cofactor in the processes of (1) transamination, (2) oxidative deamination, (3) urea cycle, (4) carbon skeleton degradation to CAC intermediates, or (5) carbon skeleton degradation to non-CAC intermediates.
Concept introduction:
Cofactors are non-protein organic compounds that are used along with the enzymes and help to carry forward the reaction. The coenzymes containing B-vitamin serve as temporary carriers of atoms or
Transamination reaction is a biochemical reaction that involves the transfer of an amino group. In transamination reaction exchange of an amino group from an
A biochemical reaction in which an
A urea cycle is a cyclic biochemical pathway that involves the production of urea using ammonium ions and aspartate molecules as nitrogen sources. The reactants in the formation of carbamoyl phosphate are ammonium ion, water, and carbon dioxide. The desired product of the urea cycle is urea.
There are 20 standard amino acids. Each amino acid has a different carbon skeleton and has a different degradation pathway for its carbon skeleton.
(a)
Answer to Problem 15.115EP
B vitamin niacin is involved as a cofactor in the process of oxidative deamination and in carbon skeleton degradation to non-CAC intermediates.
Explanation of Solution
Coenzyme nicotinamide adenine dinucleotide
Oxidative deamination reaction of glutamate requires dehydrogenase enzyme. It is an oxidoreductase enzyme and works with either
The oxidative deamination reaction of glutamate amino acid is as follows:
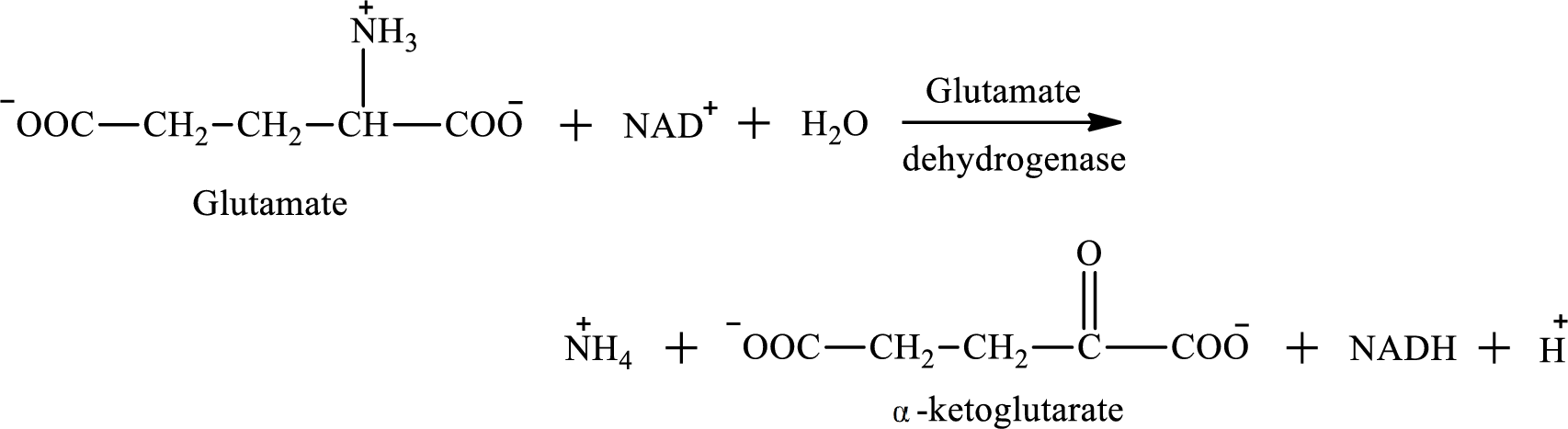
Nicotinamide adenine dinucleotide
(b)
Interpretation:
To indicate whether B vitamin folate is involved as a cofactor in the processes of (1) transamination, (2) oxidative deamination, (3) urea cycle, (4) carbon skeleton degradation to CAC intermediates, or (5) carbon skeleton degradation to non-CAC intermediates.
Concept introduction:
Cofactors are non-protein organic compounds that are used along with the enzymes and help to carry forward the reaction. The coenzymes containing B-vitamin serve as temporary carriers of atoms or functional groups in the redox and group transfer reactions associated with the metabolism of ingested food in order to obtain energy from the food.
Transamination reaction is a biochemical reaction that involves the transfer of an amino group. In transamination reaction exchange of an amino group from an
A biochemical reaction in which an
A urea cycle is a cyclic biochemical pathway that involves the production of urea using ammonium ions and aspartate molecules as nitrogen sources. The reactants in the formation of carbamoyl phosphate are ammonium ion, water, and carbon dioxide. The desired product of the urea cycle is urea.
There are 20 standard amino acids. Each amino acid has a different carbon skeleton and has a different degradation pathway for its carbon skeleton.
(b)
Answer to Problem 15.115EP
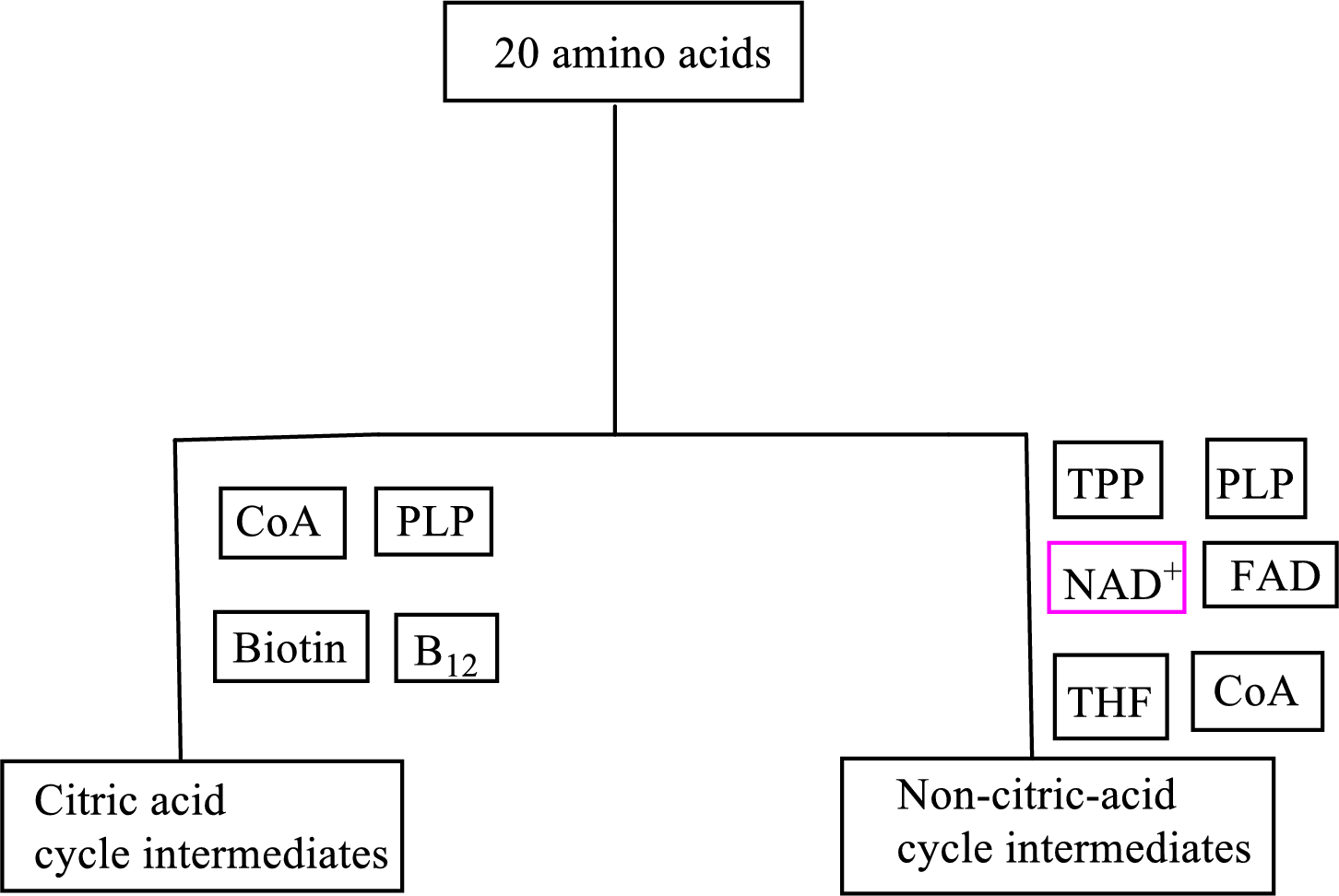
B vitamin folate is involved as a cofactor in carbon skeleton degradation to non-CAC intermediates.
Explanation of Solution
Coenzyme tetrahydrofolate
(c)
Interpretation:
To indicate whether B vitamin biotin is involved as a cofactor in the processes of (1) transamination, (2) oxidative deamination, (3) urea cycle, (4) carbon skeleton degradation to CAC intermediates, or (5) carbon skeleton degradation to non-CAC intermediates.
Concept introduction:
Cofactors are non-protein organic compounds that are used along with the enzymes and help to carry forward the reaction. The coenzymes containing B-vitamin serve as temporary carriers of atoms or functional groups in the redox and group transfer reactions associated with the metabolism of ingested food in order to obtain energy from the food.
Transamination reaction is a biochemical reaction that involves the transfer of an amino group. In transamination reaction exchange of an amino group from an
A biochemical reaction in which an
A urea cycle is a cyclic biochemical pathway that involves the production of urea using ammonium ions and aspartate molecules as nitrogen sources. The reactants in the formation of carbamoyl phosphate are ammonium ion, water, and carbon dioxide. The desired product of the urea cycle is urea.
There are 20 standard amino acids. Each amino acid has a different carbon skeleton and has a different degradation pathway for its carbon skeleton.
(c)
Answer to Problem 15.115EP
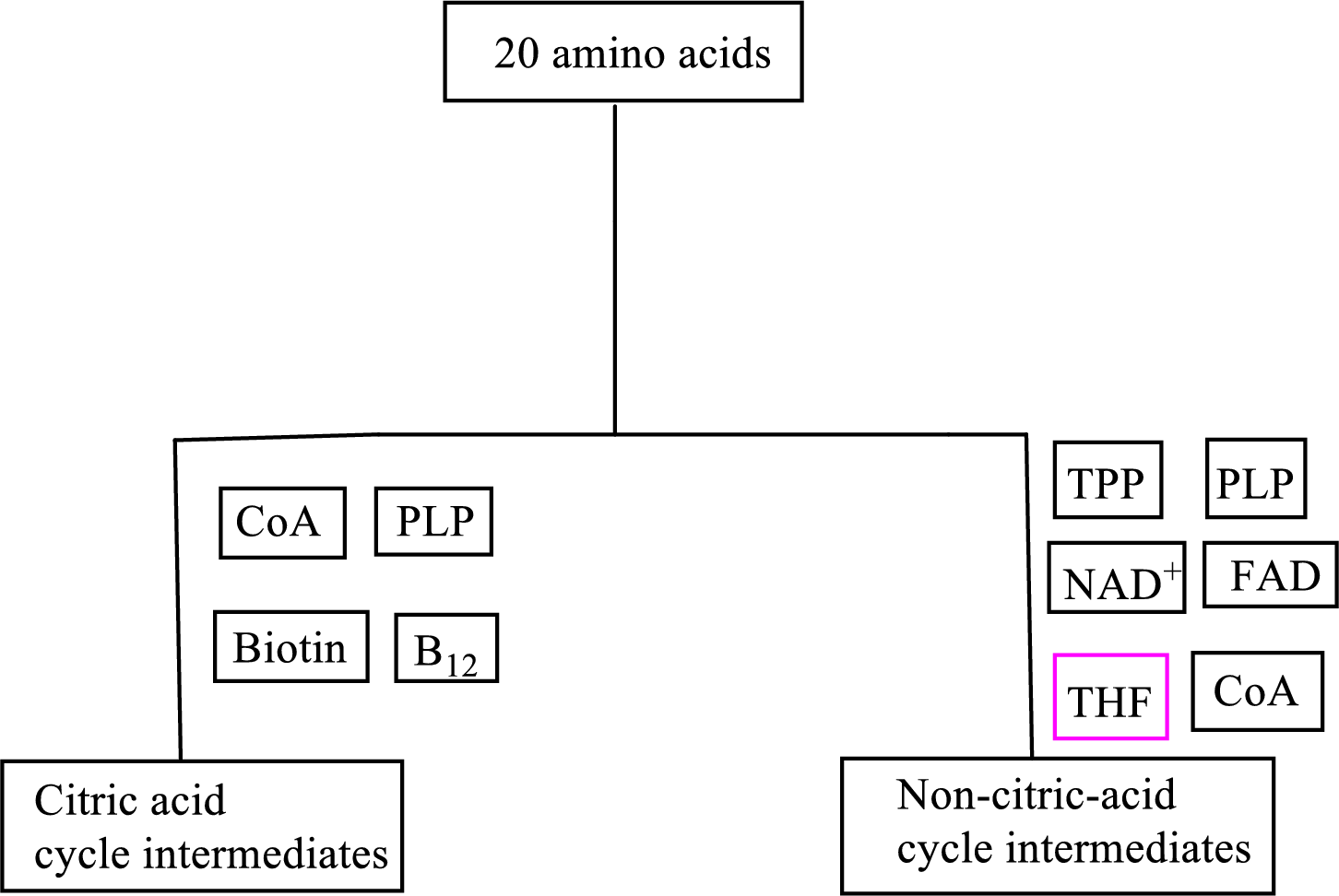
B vitamin biotin is involved as a cofactor in carbon skeleton degradation to CAC intermediates.
Explanation of Solution
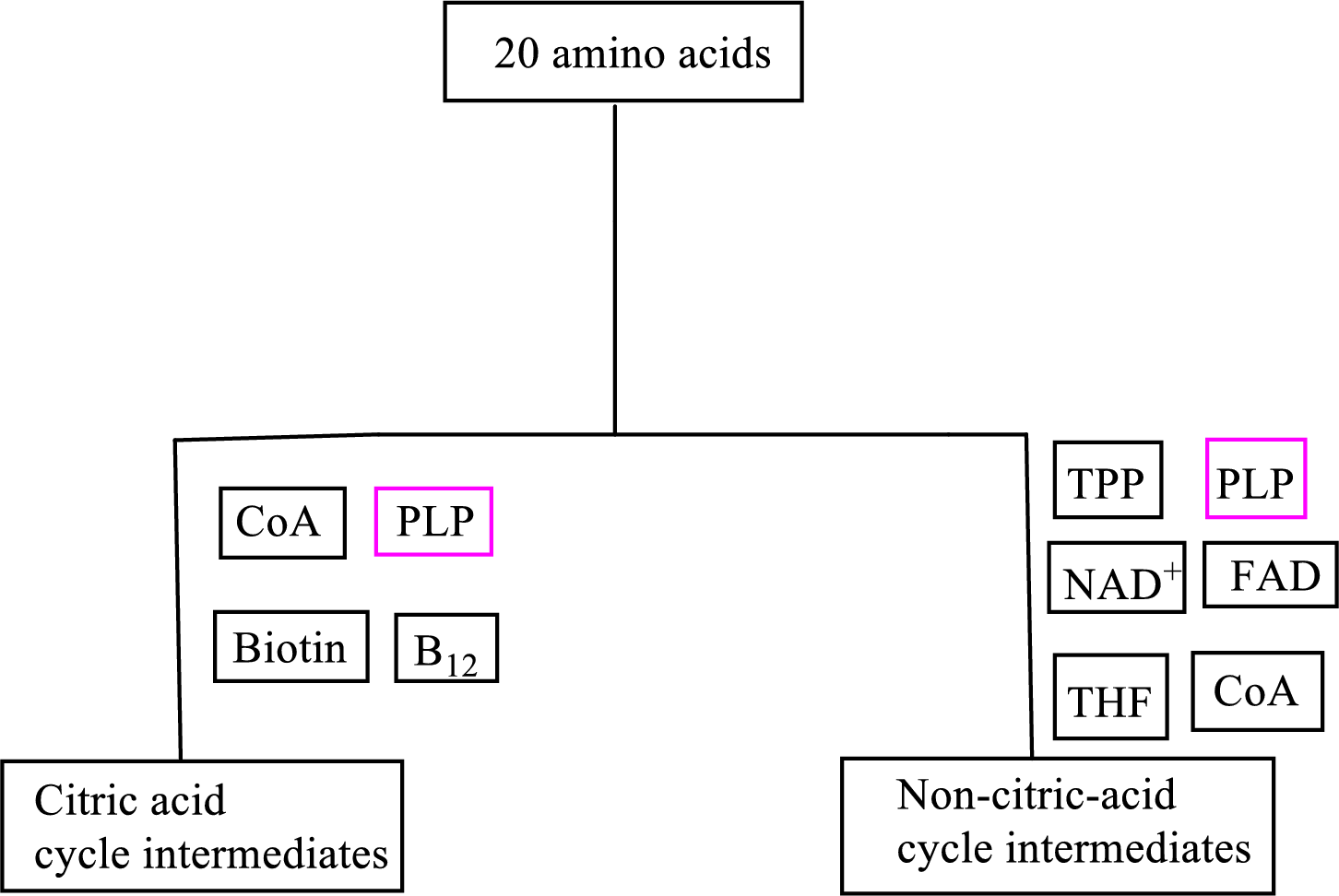
Biotin is involved in the carbon skeleton degradation pathway to CAC intermediates. An overview of the B vitamin participations in the degradation pathways for the carbon skeletons of the 20 standard amino acids is as follows:
(d)
Interpretation:
To indicate whether B vitamin
Concept introduction:
Cofactors are non-protein organic compounds that are used along with the enzymes and help to carry forward the reaction. The coenzymes containing B-vitamin serve as temporary carriers of atoms or functional groups in the redox and group transfer reactions associated with the metabolism of ingested food in order to obtain energy from the food.
Transamination reaction is a biochemical reaction that involves the transfer of an amino group. In transamination reaction exchange of an amino group from an
A biochemical reaction in which an
A urea cycle is a cyclic biochemical pathway that involves the production of urea using ammonium ions and aspartate molecules as nitrogen sources. The reactants in the formation of carbamoyl phosphate are ammonium ion, water, and carbon dioxide. The desired product of the urea cycle is urea.
There are 20 standard amino acids. Each amino acid has a different carbon skeleton and has a different degradation pathway for its carbon skeleton.
(d)
Answer to Problem 15.115EP
Explanation of Solution
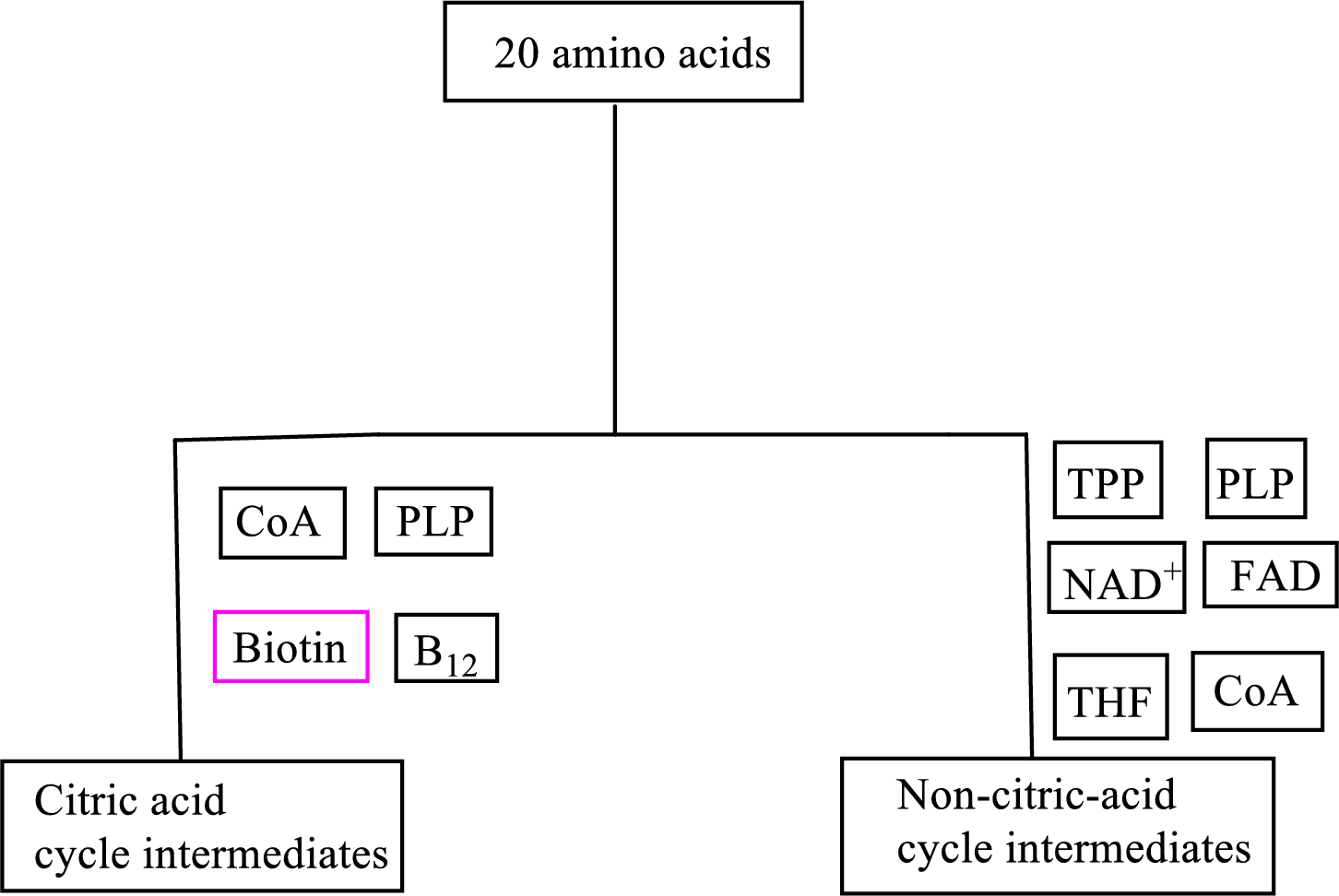
Coenzyme
Transamination reaction involves a simple transfer of amino groups but the overall reaction occurs in several steps and also requires
Coenzyme
An overview of the B vitamin participations in the degradation pathways for the carbon skeletons of the 20 standard amino acids is as follows:
Want to see more full solutions like this?
Chapter 15 Solutions
Organic And Biological Chemistry
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





