
Concept explainers
(a)
Interpretation:
The double bond in the following compound should be labeled as cis or trans.
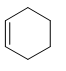
Concept Introduction:
The rotation around the carbon atoms of a double bond is restricted. So, groups on one side of the double bond cannot rotate to the other side. Depending on the side of the alkyl group attached to the double bond, there are two stereoisomers as cis and trans. Trans indicates that the alkyl groups are on opposing sides and cis indicates that the alkyl groups are on same side.
(b)
Interpretation:
The double bond in the following compound should be labeled as cis or trans.
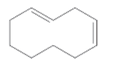
Concept Introduction:
The rotation around the carbon atoms of a double bond is restricted. So, groups on one side of the double bond cannot rotate to the other side. Depending on the side of the alkyl group attached to the double bond, there are two stereoisomers as cis and trans. Trans indicates that the alkyl groups are on opposing sides and cis indicates that the alkyl groups are on same side.
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
General, Organic, and Biological Chemistry - 4th edition
- Determine which cyclohexane structure has the MOST energy (is the LEAST stable)?arrow_forwardIt is easy to imagine a cyclohexane as a flat hexagon and a lot of the time we draw it that way. Looking at 1,3,5-triethylcyclohexane we cannot tell the stability of the molecule from looking at the flat 2D drawing. Explain why we need to look at the 3D configuration and what conformation (axial,equatorial) would each of the three ethyl groups be in for the most stable configuration.arrow_forward1. What type of reaction that all organic compounds undergo? 2. What is the total bond order of sulfur in CH3SCH3? 3. Explain the meaning in organic formulas of a pair of parentheses with no subscript behind it, such as in CH3CH2CH(CH3)C3H7arrow_forward
- Consider the molecule trans-1-ethyl-2-methylcyclohexane. What can you say about the most stable form of this molecule? O The ethyl is axial and the methyl is equatorial O The methyl is axial and the ethyl is equatorial Both substituents are axial Both substituents are equatorialarrow_forwardDraw all of the constitutional isomers with molecular formula of C6H12 that possess one pi bond. Don't include cyclic structures.arrow_forwardIn 1874, Dutch chemist Jacobus van't Hoff (1852–1911) and French chemist Joseph Le Bel (1847–1930) independently deduced that a carbon atom bonded to four atoms assumes a tetrahedral geometry. Prior to that time, it was believed that tetravalent carbons assumed a square planar geometry. One piece of evidence that can be used to support a tetrahedral geometry is the fact that molecules with the general formula CX2Y2 (where X and Y are either a hydrogen or a halogen atom) are always polar. Explain how this supports a tetrahedral geometry and rules out a square planar geometry. Square planar geometry Tetrahedral geometryarrow_forward
- How would you best describe the C-C bonds lengths in benzene relative to cyclohexane? Hypothesize why these results are observed.arrow_forwardWhen cyclopropane is treated with HI, 1-idopropane is formed. A similar type of reaction does not occur with cyclopentene or Cyclohexane. Suggest an explanation for cyclopropane’s reactivity.arrow_forward1. What is resonance contributor in which one or more atoms bears a formal change and the most stable resonance form? 2.what is the process of distributing electron pairs in a molecule? 3. Are organic molecules which are less sterically hindered more attractive than those which are more sterically hindered?arrow_forward
- The energy difference between a tert-butyl group going from equatorial to axial in a cyclohexane is 18.3 kJ/mol. When two of the carbon atoms are replaced with oxygen atoms (molecule B) the energy difference between the two chair conformations drops to 5.9 kJ/mol. Explain this difference. (Hint: Consider what makes putting groups axial unfavorable).arrow_forwardCircle all of the chiral centers (i.e., asymmetric carbon atoms) in the following compounds.arrow_forwardWhich of the following statement is false about conjugated systems? A conjugated system is more stable than an unconjugated A system. The s-trans isomer is favored over the s-cis isomer. A conjugated system has C multiple resonance structures. A conjugated system can only D contain two alkenes.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


