
Concept explainers
(a)
Interpretation:
The balanced chemical equation for the reaction of hydrogen sulfide and oxygen that results the formation of water vapor and sulfur needs to be determined.
Concept introduction:
A balanced chemical equation must represent the molecules of reactant and product in their molecular formulae.
A balanced chemical equation always has same number of all atoms at both sides of the equation and can be used for stoichiometric calculations.
(a)
Answer to Problem 59A
Explanation of Solution
The reaction of hydrogen sulfide and oxygen forms sulfur and water vapor. The balanced chemical equation can be written as:
In the balanced chemical equation, both sides have same number of H, O and S atoms.
(b)
Interpretation:
The balanced chemical equation for the reaction of hydrogen sulfide and oxygen that results the formation of water vapor and sulfur needs to be determined with the help of given color sketch. (Yellow balls = S, Blue = H, Red = O)
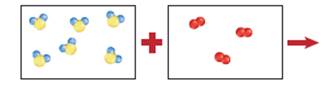
Concept introduction:
A balanced chemical equation must represent the molecules of reactant and product in their molecular formulae.
A balanced chemical equation always has same number of atoms at both sides of the equation and can be use for stoichiometric calculations.
(b)
Answer to Problem 59A
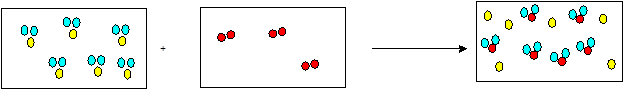
Explanation of Solution
The reaction of hydrogen sulfide and oxygen forms sulfur and water vapor. The balanced chemical equation can be written as:
In the given figure, 6 moles of H2S reacts with 3 moles of
2 moles of
So 6 moles of
Thus, all the 6 moles of
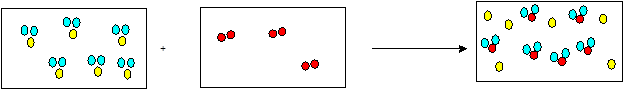
Chapter 11 Solutions
Glencoe Chemistry: Matter and Change, Student Edition
Additional Science Textbook Solutions
Essential Organic Chemistry (3rd Edition)
Introductory Chemistry (6th Edition)
Chemistry: The Central Science (13th Edition)
Chemistry: Structure and Properties (2nd Edition)
Organic Chemistry (9th Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





