
(a)
Interpretation: The IR spectra of absorption associated with the C=C bond and trans relationship vinylic H atoms for the spectral data for trans-4-methyl-2-pentene given in the figure needs to be determined.
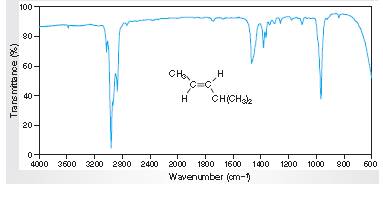
Concept Introduction:
Spectroscopy is the method of identification of structure of the organic molecules with the help of their absorption of certain
(b)
Interpretation The H1 spectra for various resonance to the hydrogen nuclei responsible for them intrans-4-methyl-2-pentene needs to be determined.
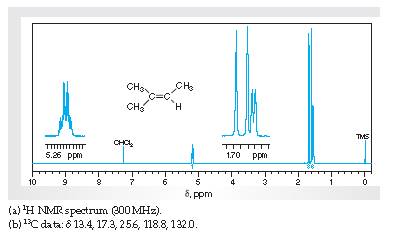
Concept Introduction:
Spectroscopy is the method of identification of structure of the organic molecules with the help of their absorption of certain electromagnetic radiations.
The H1spectroscopy or NMR is based on the nuclear magnetic resonance of radio waves on the H atoms bonded to C atoms present in the organic molecule.
(c)
InterpretationThe C13 spectra for various resonance to the C nuclei responsible for them in trans-4-methyl-2-pentene needs to be determined.
Concept Introduction:
Spectroscopy is the method of identification of structure of the organic molecules with the help of their absorption of certain electromagnetic radiations.
The H1spectroscopy or NMR is based on the nuclear magnetic resonance of radio waves on the H atoms bonded to C atoms present in the organic molecule.
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
EBK EXPERIMENTAL ORGANIC CHEMISTRY: A M
- 3. Give the structure of the product you would expect to obtain from the reaction of 2- phenylethylamine with the following reagents. a. hydrogen bromide. b. acetone. c. 1,2-epoxypropane. d. Excess methyliodide. e. sodium nitrite in dilute hydrochloric acid.arrow_forwardthis is the spectral data for 4-cyclohexene-cis-1,2-dicarboxylic acid - Write the 1H NMR spectral information in ACS formatarrow_forwarda. The structure of a standard C4 group chemically bonded to a silica surface through a silyl ether bond. b. A reasonable mechanism by which acid hydrolysis (H* and H20) might cleave the silyl ether bond, including the products of the reaction.arrow_forward
- 1. Nitrile hydrolysis to prepare carboxylic acids. a. What gas is given off in this reaction? How can it be detected chemically? b. Which spectroscopic method could be used to follow this reaction? What would you expect to see? How would you know when the reaction is complete?arrow_forwardWhich qualitative analysis test from this module can help distinguish between different kinds of ketones? Can this same test give a positive result for 4-methylacetophenone? Why?arrow_forwardWrite the reaction involved in Ferrox Test. a. What is the species responsible? b. Why is phenol negative in Ferrox Test? Based on the theoretical result, what is the order of reactivity of primary, secondary, and tertiary alcohols in the Lucas Test? a. Lucas Reagent contains ZnCl2 in HCl. What is the role of ZnCl2? What reagents are used in the esterification of Alcohols and Phenols? a. Write the reaction involved in Primary Alcohol (Ethanol) and Acetyl Chloride b. Write the reaction involved in Phenol and Acetyl Chloride What is the purpose of the Chromic acid test? a. What are the reagents used? b. Write oxidation reaction of Primary Alcohols and Secondary Alcoholsarrow_forward
- 1. Summarize the essential properties of nucleophilic acyl substitution. 2. Select one carboxylic acid that can be used in clinical diagnostic testing, nutritional research, or pharmacological studies. Give an explanation as to how the molecular property influences the bulk property of the carboxylic acid.arrow_forwardthis is the spectral data for 4-cyclohexene-cis-1,2-dicarboxylic anhydride 1. Assign the peaks in the region above 1600 cm-1 of the IR. 2. In the 1H NMR spectrum, assign the various resonances to the hydrogen nuclei responsible for them. 3. For the 13C NMR data, assign the resonances to the carbon nuclei responsible for them.arrow_forward1. In the reactions involving the three isomeric alcohols with the formula C4H9OH, describe what each of the following tests showed about reactivity of the -OH group and reactions of 1°, 2°, and 3° alcohols. • the test with neutral KMNO4 • the test with concentrated HCl 2. Predict how the fourth alcohol with the formula C4H100 would react if tested with: • 0.01 M KMNO4 • concentrated HCl at room temperature Extend Further Use your observations of the solutions formed in the previous experiments and your understanding of alcohols to complete a table like the one shown below. Research the melting and boiling points to verify your answers. Alcohol Structural Boiling point Solubility in Solubility in Color with Diagram water oil Litmus 1-Butanol 2-Butanol | 2-Methylpropanolarrow_forward
- Write a mechanism that accounts for the formation of ethyl isopropyl ether as one of the products in the following reaction. A. Write the mechanism for step one of this reaction. Show lone pairs and formal charges. Only the acidic hydrogen should be drawn out with a covalent bond. B. Write the mechanism for step two of this reaction (where the product of step one reacts with the solvent, ethanol). Show lone pairs and formal charges. Only the acidic hydrogen should be drawn out with a covalent bond. C.Write the mechanism for the last step of this reaction (formation of ethyl isopropyl ether). Show lone pairs and formal charges. Only the acidic hydrogen should be drawn out with a covalent bond. Cl- will act as the base in this reaction.arrow_forwardIn a Grignard synthesis of benzoic acid experiment, a student determines that their crude product was a mixture of benzoic acid and benzene. Describe a series of extractions using the following solvents and solutions to get pure benzoic acid or its salt in an ether solution. A. Methyl tert-butyl ether (MTBE) B. 3M HCl C. 3M NaOHarrow_forwardThe following tests or reagents could differentiate 2,3-dimethylheptane and 2,3-dimethylhepta-1,5-diene EXCEPT a. Baeyer's Test b. Br2 in water under dark conditions c. Friedel-Crafts Alkylation Test d. Combustion The following statements about anthracene are true EXCEPT: a. It gives a negative result in Baeyer's test b. It produces a yellow, smoky, sooty flame c. It is insoluble in cold H2SO4. d. It reacts with bromine under dark conditions.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





