
Concept explainers
(a)
Interpretation: The structure corresponding to the given
Concept introduction: The systematic naming of organic compound is given by
Rules for writing structural formula from IUPAC are:
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
The use of prefix
Answer to Problem 10.39P
The structure corresponding to
Explanation of Solution
The given IUPAC name is
Rules for writing structural formula from IUPAC are
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
The use of prefix
The given name is
Thus, the correct structure of
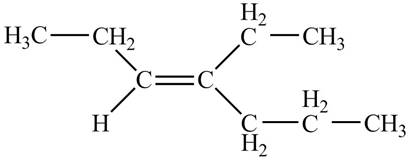
Figure 1
The structure corresponding to
(b)
Interpretation: The structure corresponding to the given
Concept introduction: The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing structural formula from IUPAC are:
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
Answer to Problem 10.39P
The structure corresponding to
Explanation of Solution
Rules for writing structural formula from IUPAC are
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
The given name is
Thus, the correct structure of
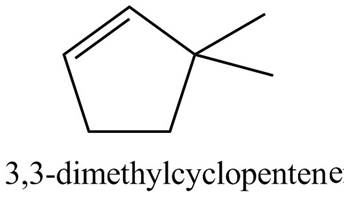
Figure 2
The structure corresponding to
(c)
Interpretation: The structure corresponding to
Concept introduction: The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing structural formula from IUPAC are:
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
Answer to Problem 10.39P
The structure corresponding to
Explanation of Solution
Rules for writing structural formula from IUPAC are
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
The given name is
Thus, the correct structure of
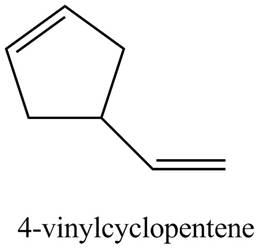
Figure 3
The structure corresponding to
(d)
Interpretation: The structure corresponding to
Concept introduction: The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing structural formula from IUPAC are:
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
The use of prefix
Answer to Problem 10.39P
The structure corresponding to
Explanation of Solution
Rules for writing structural formula from IUPAC are
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
The use of prefix
The given name is
Thus, the correct structure of
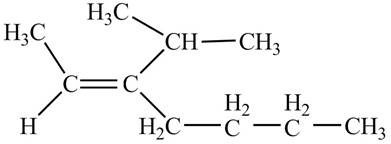
Figure 4
The structure corresponding to
(e)
Interpretation: The structure corresponding to
Concept introduction: The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing structural formula from IUPAC are:
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
Answer to Problem 10.39P
The structure corresponding to
Explanation of Solution
Rules for writing structural formula from IUPAC are
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
The given name is
Thus, the two correct structure of
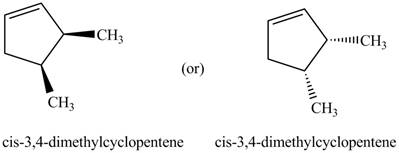
Figure 5
The structure corresponding to
(f)
Interpretation: The structure corresponding to
Concept introduction: The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing structural formula from IUPAC are:
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
Answer to Problem 10.39P
The structure corresponding to
Explanation of Solution
Rules for writing structural formula from IUPAC are
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
The given name is
Thus, the correct structure of
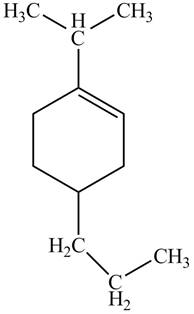
Figure 6
The structure corresponding to
(g)
Interpretation: The structure corresponding to
Concept introduction: The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing structural formula from IUPAC are:
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
Answer to Problem 10.39P
The structure corresponding to
Explanation of Solution
Rules for writing structural formula from IUPAC are
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
The given name is
Thus, the correct structure of
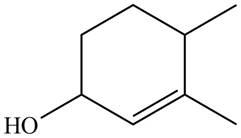
Figure 7
The structure corresponding to
(h)
Interpretation: The structure corresponding to
Concept introduction: The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Rules for writing structural formula from IUPAC are:
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
Answer to Problem 10.39P
The structure corresponding to
Explanation of Solution
Rules for writing structural formula from IUPAC are
1. First identify the word root for the given compound.
2. The suffix used in the compound like –ene.
3. Identify the position, location, and number of the substituent bonded to the carbon chain.
The given name is
Thus, the correct structure of
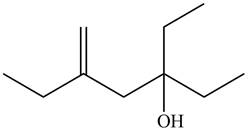
Figure 8
The structure corresponding to
Want to see more full solutions like this?
Chapter 10 Solutions
Organic Chemistry
- 1. Draw the structure for each compound. a.(3R)-3-methylhexane b. (3R,5S,6R)-5-ethyl-3,6-dimethylnonanearrow_forwardII. Draw the structures corresponding to each name. 1. m-bromoaniline 2. p-bromotoluene 3. 1-chloro-3,5-dimethylbenzene 4. O-iodophenol 5. 1,3,5-trichlorobenzene 6. O-hydroxybenzenesulfunic acidarrow_forwardH3C. CH3 What is the systematic IUPAC name of this compound? H;C Select one: a. 1-(3-isopropylcyclohexene-6-yl)ethane b. 3-ethyl-6-(propan-2-yl)cyclohex-1-ene O c. 6-ethyl-3-isopropylcyclohex-1-ene d. 2-(3-ethylcyclohexene-6-yl)propane e. 3-ethyl-6-isopropylcyclohex-1-enearrow_forward
- C. Draw structures in blank boxes to complete the following reactions. cyclopentanone 5-hydroxy-3- methylpentan-2-one & HO H HO OH cat. HCI H₂O 1. H₂NNH₂, EtOH 2. NaOH Zn(Hg), H3O+ NaBH3CN, EtOH, pH 5 cat. HCI cat. HCI pH 5arrow_forwardC. Give the structure corresponding to each name. 1. 3-chloro-2-methylhexane 2. 4-ethyl-5-iodo-2-methylbenzene 3. cis-1,3-dichlorocyclopentane 4. 1,3,5-tribromobenzene 5. propyl chloride 6. sec-butyl bromidearrow_forwardWhich structure is trans-3-hexene? CH3. H CH3, H OCH3CH₂ H OCH3CH₂ H C. C. FC. C H CH₂CH₂CH3 CH₂CH₂CH3 H H CH₂CH3 CH₂CH3 Harrow_forward
- Give the structure corresponding to each name. a. 3-chloro-2-methylhexane b. 4-ethyl-5-iodo-2,2-dimethyloctane c. cis-1,3-dichlorocyclopentane d. 1,1,3-tribromocyclohexane e. propyl chloride f. sec-butyl bromidearrow_forward1. Using IUPAC guidelines, name the following organic compounds. Answerarrow_forward8. What is the IUPAC name of CHCH, CH, CH3-CH-O-CH, CH; Da. 1-ethoxy-1-methylbutane Ob. 2-ethoxypentane O c. 1-methoxyhexane Od. 2-ethoxy-2-n-propylethanearrow_forward
- Name each alkyne. a. CH=C-CH-CH3 CH3 b. CH3-C=C-CH-CH-CH,-CH, CH3 CH3 c. CHEC-C-CH,-CH3 CH2 ČH, CH3 d. CH;-C=C-CH-C-CH, CH2 CH3 ČH3arrow_forwardName each alkyne. CH; c. CHEC-Ç-CH2-CH3 CH2 CH3 CH; d. CH3-C=C-CH--CH3arrow_forwardCHCHS CH, CH CH CHCH, A D Which is the most stable alkene? Choose. Which is the least stable alkene? Choose.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





