
Concept explainers
(a)
Interpretation:
The alcohol from which the compound
Concept introduction:
Primary and secondary alcohols can be oxidized into
Answer to Problem 10.31P
The
Explanation of Solution
Primary and secondary alcohols can be oxidized into aldehydes and ketones using
The compound
The carboxylic acid,
The alcohol from which the compound
(b)
Interpretation:
The alcohol from which the compound
Concept introduction:
Primary and secondary alcohols can be oxidized into aldehydes and ketones using
Answer to Problem 10.31P
The
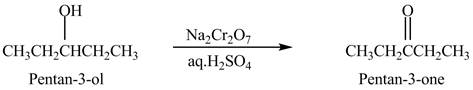
Explanation of Solution
Primary and secondary alcohols are oxidized into aldehydes and ketones using
The secondary alcohols are converted into ketones on oxidation.
The compound
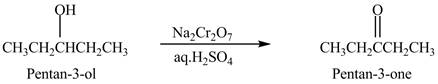
Figure 1
The ketone
The alcohol from which the compound
(c)
Interpretation:
The alcohol from which the compound
Concept introduction:
Primary and secondary alcohols can be oxidized into aldehydes and ketones using
Answer to Problem 10.31P
The
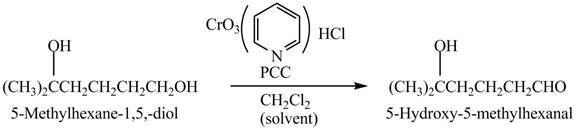
Explanation of Solution
Primary and secondary alcohols can be oxidized into aldehydes and ketones using
The primary alcohols are converted into aldehydes under anhydrous conditions.
The compound

Figure 2
The aldehyde,
The alcohol from which the compound
(d)
Interpretation:
The alcohol from which the compound
Concept introduction:
Primary and secondary alcohols can be oxidized into aldehydes and ketones using
Answer to Problem 10.31P
The
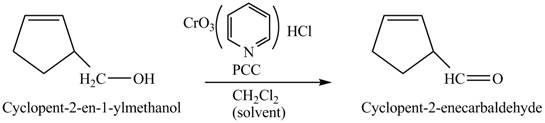
Explanation of Solution
Primary and secondary alcohols can be oxidized into aldehydes and ketones using
The primary alcohols are oxidized into aldehydes under anhydrous conditions.
The compound
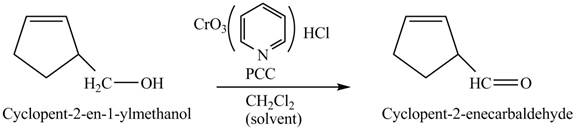
Figure 3
The compound
The alcohol from which the compound
Want to see more full solutions like this?
Chapter 10 Solutions
Organic Chemistry Study Guide and Solutions
- Draw the structures of the following compounds:(a) Ethanoic acid(b) Bromopentane(c) Butanonearrow_forwardGive simple chemical tests to distinguish between the following pairs of compounds:(i) Ethanol and Phenol(ii) Propanol and 2-methylpropan-2-olarrow_forwardWhat is the chemical equation when nitrating mixture is added to the following:(a) Cyclohexane (b) Cyclohexene(c) Toluenearrow_forward
- What is the major organic product of the following reaction? (a) (b) NaBH4 CH3CH₂OH ? (c) (d) OHarrow_forward1,4-Dioxane is made commercially by the acid-catalyzed condensation of an alcohol.(a) Show what alcohol will undergo condensation, with loss of water, to give 1,4-dioxane.arrow_forwardUse the symbol (V) to indicate a positive reaction and the symbol (X) to indicate a negative reaction and a () if a test was not performed in the shaded columns. Don't forget to write the number of your unknown underneath where it says number! IDENTIFICATION OF A CARBONYL COMPOUND (GROUP I) Unknowns Number Number methyl ketone Test Aldehyde ketone DNP Fehling's Tollen's X lodoform X Therefore, unknown number is IDENTIFICATION OF ALCOHOL (GROUP II) Unknown Primary alcohol Secondary Tertiary alcohol number: alcohol Test Lucas (in ~5 mins) (immediately) Bordwell - Wellman Therefore, unknown number is > x x >arrow_forward
- 6) Which is the organic product for the following reaction? (a) (b) (c) (d) сон COOH ОН ОН COOH COOH KMnO4 H2Oarrow_forwardWhich of the isomeric C4H₁0O alcohols can be prepared by hydrogenation of aldehydes? Which can be prepared by hydrogenation of ketones? Which cannot be prepared by hydrogenation of a carbonyl compound?arrow_forwardPredict which member of each group is most soluble in water, and explain the reasons for your predictions.(a) butan-1-ol, pentan-1-ol, or propan-2-ol(b) chlorocyclohexane, cyclohexanol, or cyclohexane-1,2-diol(c) phenol, cyclohexanol, or 4-methylcyclohexanolarrow_forward
- Draw the structure of C4H1,O if the compound: (1) reacts with Na but fails to react with a strong oxidizing agent such as K,Cr,O;; (2) gives a negative iodoform test; and (3) gives a positive Lucas test in 4 minutes. (ii) Give structures and IUPAC names of the alcohols formed from (CH3),CHCH=CH2 by: (i) dilute H,SO,; (ii) В,Нь, then H,Oz, он.arrow_forwardGive an IUPAC and common name for each of the following naturally occurring carboxylic acids: (a) CH3CH(OH)CO2H (lactic acid); (b) HOCH2CH2C(OH)(CH3)CH2CO2H (mevalonic acid).arrow_forward4 But-2-enal, CH₂CH=CHCHO, is a pale yellow, flammable liquid with an irritating odour. (a) But-2-enal exists as two stereoisomers. Draw skeletal formulae to show the structure of the two stereoisomers of but-2-enal. (b) (i) Describe a simple chemical test that would show that but-2-enal is an aldehyde. (ii) Explain why this test gives a different result with aldehydes than it does with keton (c) But-2-enal also reacts with sodium borohydride, NaBH4. (i) Identify the organic compound formed in this reaction. (ii) State the type of chemical reaction occurring. (d) Precautions must be taken to prevent but-2-enal catching fire. Construct a balanced equation for the complete combustion of but-2-enal, C₂HO.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





