Q: Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product…
A: Curved arrows show the flow of electrons from electron rich species (species containing loan pair or…
Q: (S)-2-butanol reacts with potassium dichromate (K2CrO4) in aqueous sulfuric acid to give A (C4H8O).…
A: In the first step of the reaction (S)-2-butanol reacts with potassium dichromate (K2CrO4) in aqueous…
Q: Br DBN > Ez Reaction
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: 9) Propose an efficient synthesis that completes this transformation ΟΗ OH
A:
Q: 4. Predict major product for the following reactions: a. OH CrO3 H3O+ Br 1. NaCN b. 2. H3O+ Br C. 1.…
A: Note:“Since you have posted a question with multiple sub parts, we will provide the solution only to…
Q: For the reaction below: 1. Draw all reasonable elimination products to the right of the arrow. 2. In…
A: Alkyl halides undergo elimination reactions via two mechanisms. One is E1, and the another is E2…
Q: There are TWO reactions that will happen when we react the molecule shown below with methanol under…
A: Trans-esterification:When ester in treated with excess alcohol in presence of acid catalyst, a new…
Q: rand name of soft drink oncentration of undiluted standard phosphate solution, M 3x/03 M Solution…
A: The objective of this question is to calculate the concentration of phosphoric acid in the undiluted…
Q: Write an acceptable IUPAC name for the compound below. (Only systematic names, not common names are…
A: Organic compounds can be defined as compounds that contain carbon and hydrogen atoms. We have been…
Q: Check the box next to each molecule on the right that has the shape of the model molecule on the…
A: Since the central atom has four bonded atoms or groups of atoms attached to it, it confirms the…
Q: 6E. Estimate the concentration of oxalic acid (HO2CCO2H) of 0.001M dipotassium oxalate (K+ O2C−CO2-…
A:
Q: If all of the electron groups around a single central atom are bonding, and the same outer atom is…
A: All of the electron groups around a single central atom are bonding, and the same outer atom is…
Q: Write a mechanism to account for the following transformation. + &.r NaOH Δ
A:
Q: Indicate the mechanism of Hofmann degradation of the compound N-(2-phenylethyl)butan-1-amine.…
A: The objective of the question is to understand the mechanism of Hofmann degradation of the compound…
Q: A buffer solution is 0.444 M in and 0.251 M in . If for is , what is the pH of this buffer…
A: I was provided the solution for this problem, I hope it will helps you. Explanation:Step 1:
Q: 5) Suggest a series of reagents that could be used to carry out the following transformation. More…
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: This energy diagram shows the allowed energy levels of an electron in a certain atom or molecule: D…
A: Answer:In an atom, electron can be at different energy levels and each energy level has certain…
Q: a species that is oxidized is also the oxidizing agent? True or false
A: The objective of the question is to understand the relationship between a species that is oxidized…
Q: Which of the following is an important reactive electrophile in aromatic ring nitration? ONO2(+) O…
A: We have to find electrophile in aromatic ring nitration.
Q: The following reactions involve combinations of pericyclic reactions. Give detailed mechanisms for…
A: the objective of this question is to To create a cyclohexene ring, a conjugated diene, and a…
Q: Hydrazine, N,H,, reacts with oxygen to form nitrogen gas and water. N₂H(aq) + O2(g) → N2(g) +…
A: mass of N2H4 = 2.25 gmolar mass of N2H4 = 32.04 g/molVolume of N2 = 0.950 LT = 295 KP = 1.00 atmR =…
Q: ? O H₂O, HCI, benzene 1. HCI, 2. ONa
A: The given scheme involves the conversion of alkene to esters. Scheme for the conversion of alkene to…
Q: The following transformation will require the use of a blocking group. H₂N. True O False NH2 H₂N NO₂…
A: Given statement: The following transformation will require the use of a blocking group.We have to…
Q: O₂N. H₂SO4 (fuming)
A: The objective of this question is to find out the major product of the given organic reaction with a…
Q: Consider the following reaction at 298K. Fe2+ (aq) + Ni →→ Fe (s) + Ni²+ (aq) Which of the following…
A: The objective of the question is to determine which of the given statements are correct for the…
Q: The elemental analysis of an organic solid extracted from gum arabic (a gummy substance used in…
A: “Since you have posted multiple questions, we will provide the solution only to the first question…
Q: 2) Draw the isobaric phase diagram at 1 atm of the following two-component liquid system: water…
A: The isobaric phase diagram shows the % weight of each component in the mixture in the liquid phase…
Q: 2. At 298.15 K, a reaction's equilibrium constant is 0.497. The equilibrium constant becomes 1.93…
A: The objective of this question is to calculate the standard enthalpy change (ΔH°) and the standard…
Q: 18. Draw a 3D representation of the aromatic heterocycle below showing the orbitals and electrons…
A: The given molecule is N-methyl pyrrole. The compound has 4-pi electrons and one lone pair in…
Q: Draw the structural formula for all the alkenes with the indicated molecular formula that, without…
A: Bromonydrin formation is addition reaction of Br and OH across the alkene and the product is called…
Q: Alkyl halides undergo nucleophilic substitution and elimination reactions. When the kinetics of the…
A: The given mechanism follows E2 mechanism.Explanation:
Q: What are the primary organic products of hydrolysis of the following molecule?
A:
Q: Write the acidic equilibrium equation for HPO 2. Be sure to include the proper phases for all…
A: Given,The polyatomic ion is:HPO42-
Q: Curved arrows are used to illustrate the flow of electrons. Use the reaction conditions provided and…
A: The objective of the question is to predict the product formed in the following reaction…
Q: Using the table of indicators identify which of the given indicators would be appropriate for the…
A: Answer:Indicator is the chemical substance that indicates the completion of titration by changing…
Q: Macmillan Learning Complete the reaction. Add hydrogen atoms and charges to the appropriate atoms.…
A: The compounds having -CONR2 group where R can be alkyl groups or H atoms are identified as amides.…
Q: Which among the conformers below is the most stable conformation for cis-1-bromo-3…
A: In a stable chair conformation, the bulky groups occupy equatorial positions rather than axial…
Q: M of NaOH: 0.100 Volume of HCI 25mL = 0.025L Moles of HCI = 2.5 mol What is the Volume of NaOH…
A: Since you have posted a question with multiple sub-parts, we will provide the solution only for the…
Q: Tag each carbon atom in the main chain of the 2-methylbutane (C,H12) molecule drawn below. H H…
A: Given,The structure of the molecule 2-methylbutane ( C5H12 ) is:
Q: Predict the major product of the following reaction. A OH IV H₂O H2SO4 OH ווו OH £ IV
A: Alkenes are compounds that have double bonds so they have excess pi electron density. Thus they are…
Q: 5. Describe another technique you could use to separate vanillin and ortho-vanillin. Draw a diagram…
A: It is required to separate vanilin and ortho-vanilin
Q: is the formula of h20
A: To find the formula for h20
Q: Consider the titration of 25.00 mL of 0.200 M methylamine (CH3NH2). The titrant is 0.120 M HCI.…
A:
Q: Draw the structure of the organic reactant, and write the chemical formula of the reagent used to…
A: Given is organic reaction.The given product is 2,6-dinitroanisole.So, starting compound of this…
Q: A student ran the following reaction in the laboratory at 698 K:2HI(g) H2(g) + I2(g)When he…
A: The objective of the question is to calculate the equilibrium constant, Kp, for the given reaction.…
Q: Synthesize O Br =
A:
Q: 3) Draw the major product expected for the reaction below: be HCI (cat.) CH3CH2OH
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: Consider the hypothetical thermochemical equation 3 A + B → 2 C for which ΔH = 58.7 kJ/mol. If the…
A: The objective of the question is to find out the quantity in moles of C that would be produced when…
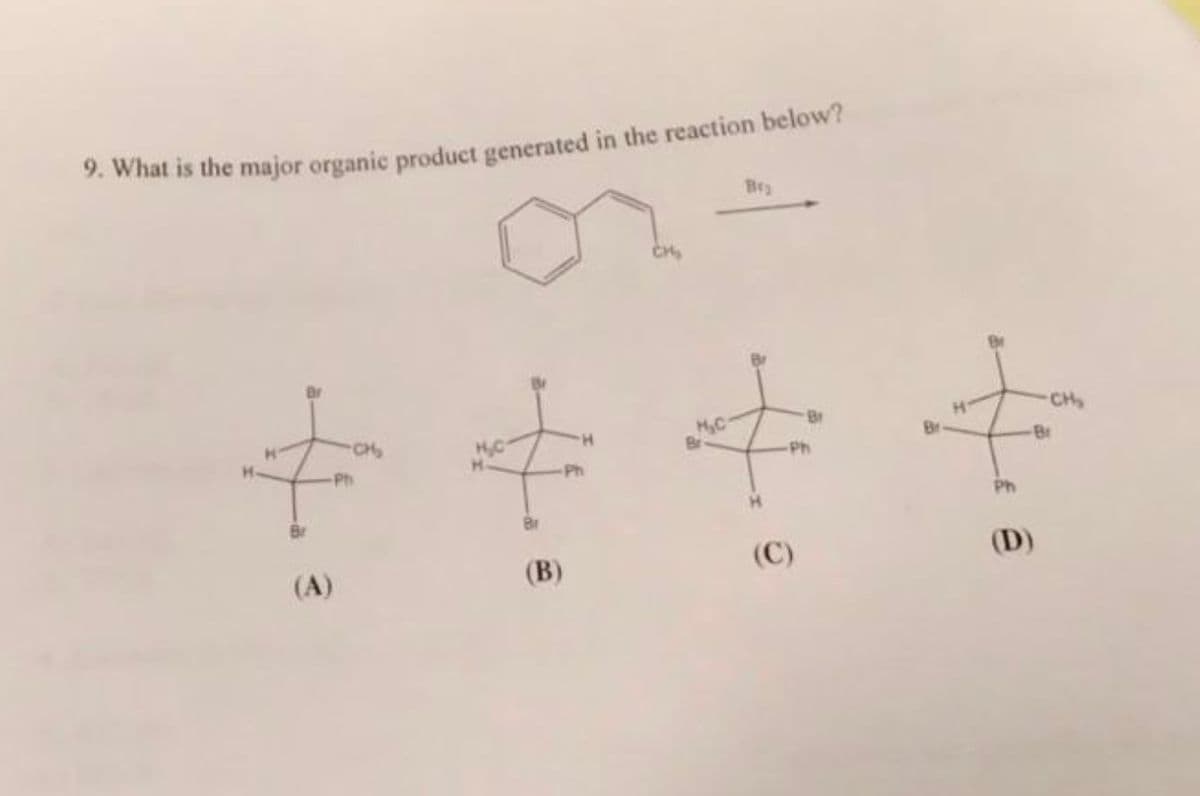
Q: Draw the structure(s) of the major organic product(s) of the following reaction. +4 CrO3/aqueous…
A: We have to predict the product.
Q: Oraw the curved arrows for the following step (mechanism patterns). (Take it one step at a time and…
A: The objective of the question is to draw the curved arrows for the given mechanism in which…
Step by step
Solved in 3 steps with 3 images

- Given the following reaction sequence: What are the reagents for reaction 3? A) NaNH2 excess followed by water B) sulfuric acid and heatWhat are the names of the compound A, B, C, D, E, F, G & H. Hence suggest structures for compound C and E. What are the reagents and reaction conditions used for the following conversions? (i), (ii), (iii), (iv) & (v)Which of the following compounds produce the 3 products shown on reaction with KMnO4 and acid (H+). a. I b. II c. III d. II and III
- Suggest an explanation for the fact that the order of reactivity of the halides toward n-butylbrosylate in acetone is Cl” > Br” > I~ when (C4H9)4N* is the cation of the halide salt butI” > Br” > Cl” when Li’ is the cationWhich of the following reagents will not undergo a substitution reaction with the benzenediazonium chloride salt. A. H3PO3, H2O B. CuCN C. Cu2O, Cu+2, H2O D. H3PO2 , H2O E. KIFor each of the following mixtures of reactants, give (i) a plausible chemical equation and (ii) structurefor the organometallic product, and (iii) general reason for the course of the reaction: (a)methyllithium and W(CO)6, (b) Co2(CO)8 and AlBr3.
- 1. What type of reaction explains solubility of phenol in sodium hydroxide? Explain the organic product formed. 2. How do the reactions of phenol samples with FeCl3 compare? Which structural component of the phenols account for the observation3. Predict the products of the reaction of the following substances with CrO3 inaqueous acid:Reaction of Aldehydes and Ketones with Organometallics: Grignard and Lithium For the following reactions: 1) Illustrate the reaction for the formation of the organometallic 2) Write the product of the reaction 3) Illustrate the mechanism of the reaction with arrow movement.

