Q: Indicate whether the salts below would form solutions that are acidic, basic, or neutral in pH. KCIO...
A:
Q: 702.0mc 3. a) How many mLs of 0.600 M calcium chloride are needed to provide 0.110 moles of chloride...
A: 3.a) We have to calculate the amount of 0.600 M of calcium chloride required to produce 0.110 moles ...
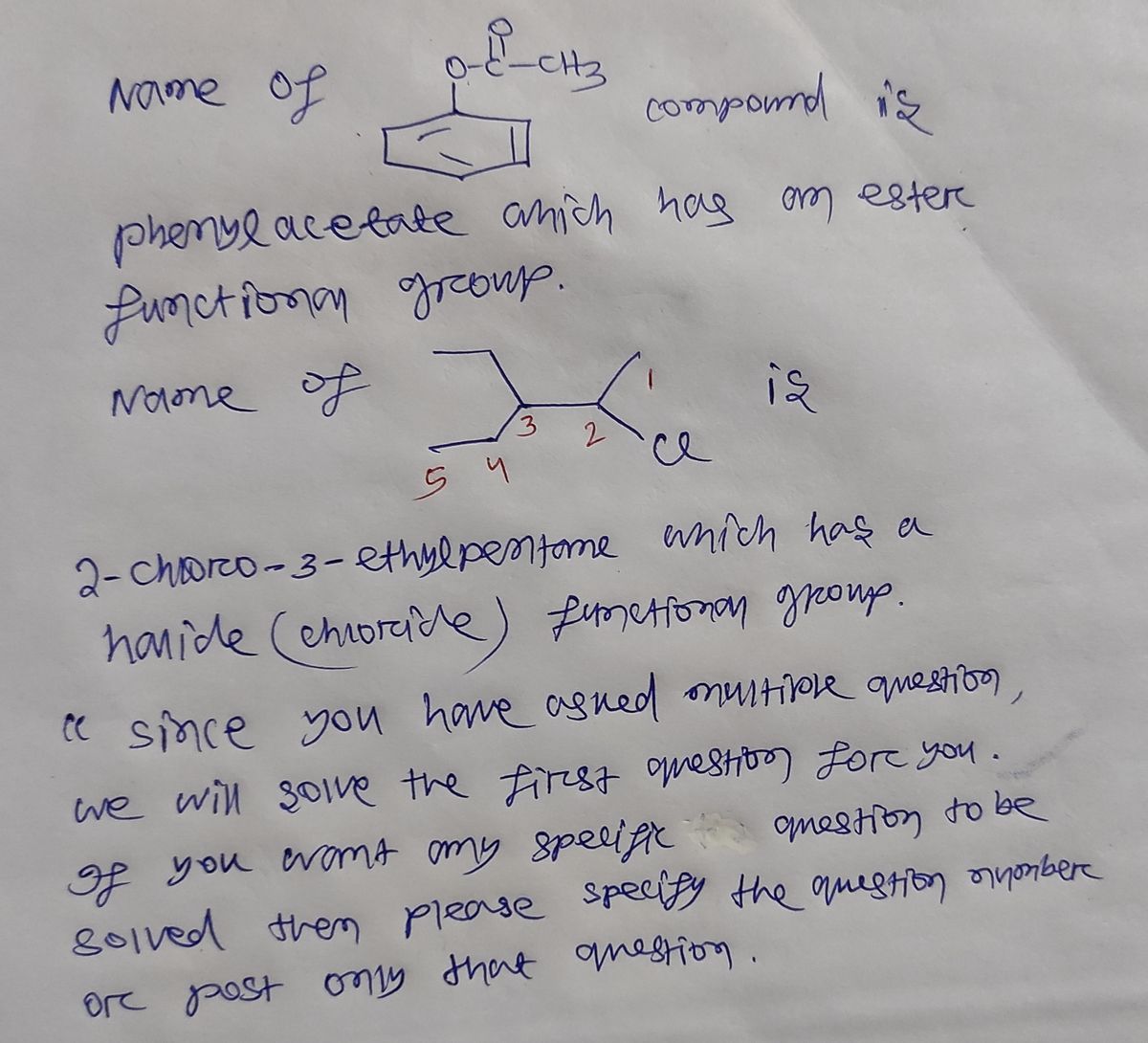
Q: what are the structures for the dollowing written out compltetly b) hexonoate reacts with methyl h...
A:
Q: milliliters
A:
Q: the rate constant for the first-order decomposition of N2O is 2.97 s^-1. what s the half-life of the...
A:
Q: What mass of CO is required to react with 25.13 g of F*e_{2} * O_{3} according to the equation : Fe ...
A:
Q: Part B: NH2
A: A question based on molecules that is to be accomplished.
Q: According to the Evan's pKa table, phenol has a pKa of 9.95. p-Nitro and m-nitrophenol have pKas of ...
A:
Q: large containers of sulfuric acid solution, 9. А chemist has two with different concentrations of ac...
A: Soln
Q: Solving an enthalpy of neutralization problem. (see Chemistry 2e, Example 5.5): when 47.35 g of wate...
A: Given: Mass of water = 47.35 g. Initial temperature of water = 22.6 oC And final temperature of wate...
Q: Calculate the pH of a 2.50x10-4M solution of hydroiodic acid, HI. pH =
A:
Q: A side reaction in the manufacture of rayon from wood pulp is 3CS2 + 6NAOH → 2NA2CS3 + Na2CO3 + 3H2O...
A: The balanced reaction given is, Given: Moles of NaOH taken = 2.78 mol. Volume of CS2 taken = 92.5 m...
Q: 3. What is the pH of a cleaning solution with a [H,O'] = 7.4 x 10 M H,O? ibility Good to go
A: Formula to calculate pH is :- pH = -log[H3O+]
Q: S, For ench renction. A7 write n word erntin B) Write he balmced olecuir equton C) write Hhe lonlmce...
A: 1) The reactants given are AgNO3 (aq) i.e. silver nitrate and NaOH (aq) i.e. sodium hydroxide.
Q: 6. A solution is made by mixing 100.0 mL of 0.200 M calcium chloride with 50.00 mL of 0.200 M sodium...
A: Soln
Q: After the reaction is complete, 6.22 g of nitrogen oxide are obtained. write the balanced equa...
A: Here 7.6 g NH3 is mixed with 10 g of O2 to produce 6.22 g of NO experimentally in Ostwald process. W...
Q: hy is bromination more selective than chlorination?
A: bromination more selective than chlorination because
Q: Determine whether each of the molecules is aromatic, nonaromatic, or antiaromatic.
A: Aromatic compounds are made up of conjugated planar ring systems with delocalized pi-electron clouds...
Q: milliliters
A:
Q: onvert each of the following pH values to a hydroxide ion concentration. y = 1.0E-14. 1.32 i M eText...
A: pH = -log [H+] [H+] x [OH-] = 1 x 10-14 As per our guidelines we can only solve first three sub-part...
Q: The equilibrium constant, Ke, for the following reaction is 1.80x104 at 298 K. NH4HS(s) =NH3(g) + H2...
A: Given, NH4HS(s) ⇌ NH3(g) + H2S(g) Temperature (T) = 298 K Equilibrium constant (KC) = 1.80 × 10-4 In...
Q: Why is the existence of a series of entering groups with different rate constants evidence for an as...
A: Introduction : Ligand are those which give their lone pair of electron to the central metal atom or...
Q: unit 3 Test - Energy in Which statement about the change in bond energy of this reaction is correct?...
A: Reaction :- CH4 + 4Cl2 ------> CCl4 + 4HCl
Q: Which is statement is true about the neutrons of atom?
A: (a) Option (a) is incorrect. Atomic mass equals the sum of neutrons and protons present in the n...
Q: What is the formula fortheedges, expressed as “f”, if the radius of the atom is expressed as “k”: a...
A: In general, edge length is represented by 'a' and radius is represented by 'r' Here , radius is den...
Q: Which species is the conjugate base of HPO4 2-? Write the formula net charge
A: A base is any substance that can accept proton from any other substances or substance that ionize in...
Q: 10. Which of the following describes a crystalline solid? a. Crystalline solids have particles that ...
A: There are three phases of matter. The phase of matter can be interchanged by changing the temperatur...
Q: A mixture of 0.02993 mol of O, 0.02165 mal of NOCI, 0.02691 mol of NO, and 0.01037 mol of C is place...
A: Answer: First of all, with the help of values of initial moles of gases and equilibrium moles of chl...
Q: What is a mixture is a combination of
A: To explain: A mixture is a combination of what?
Q: The chemical reaction shown was performed and the concentration of CCl4CCl4 was measured over time. ...
A:
Q: Determine if the statements are true. The larger the surface area, the higher the dissolution. ...
A: True or false: As per our guideline we have to answer first three questions only:
Q: a. Mg I Mgaa) l Cuag) I Cus) b. Nice I Niaa) | Agiaa) I Ag) 3. For the cell notations in the previou...
A: Here from the given Galvanic cell notation we have to write the cell reaction. For this we have to w...
Q: Fluoride ion is added to drinking water to prevent tooth decay. What is the mass in grams of sodium ...
A: Given Formula units = 1.19 × 1019
Q: c. 20.45 g Al(NOsla 5. A saline solution with a mass of 233 g has 34.5 g NaCl dissolved in it. What ...
A:
Q: (PP-1 and PP-2). PP-1 has Mw of 100,000 g/mol and PP-2 has Mw 500,000 g/mol, If the PDI (Molecular w...
A: The correct option is:
Q: Calculate the pH of a solution that is 0.0400 M in a) NaH,PO. b) NaHC:O. c) NaHS
A: Soln
Q: Why do meltiing, freezing and boiling point varies and depends on a kind of matter? Not less than 6 ...
A: Matter can be defined as substance that possess definite mass and volume. Matter can be solid, liqui...
Q: No of D isomers are possible for hexose Select one: a. 8 b. 4 c. 6 d. 16
A:
Q: 3) Design a reasonable synthesis for the following transformation. = trort + B.
A: Here we have synthesize the given polymer which is a polyester obtained from starting material 1,4-d...
Q: According to the following reaction, how many grams of calcium oxide will be formed upon the complet...
A:
Q: KHP
A:
Q: identify each substance Acid Base conjugate acid Conjugate base
A:
Q: Differentiate intensive to extensive properties. Not less than 6 sentences.
A:
Q: 4. [4] Give a valid set of four quantum numbers for each electron in the valence sh ell of a ground ...
A: There are four set of quantum numbers which describes or gives all the information about an electron...
Q: Element X has two isotopes. If 72.0% of the element has an atomic mass of 84.9 atomic mass units, an...
A: Given : We have to calculate the average atomic mass.
Q: Provide the missing reagents required for the following transformation: OH OH
A: Alkene reacts with peracid, RCO3H (such as metachloroperbenzoic acid, mCPBA) to form an epoxide. The...
Q: . Which graph correctly displays the relationship between wavelength, in meters, and frequency, in h...
A: To explain: The graphical relationship between the wavelength in m and frequency in the hertz of the...
Q: 3. Consider Lewis structure for hydrogen fluoride (HF). a. If hydrogen fluoride were to behave as an...
A: An acid donates proton while a base accepts proton. HA---> H+ + A- HA is acid while A- is con...
Q: a) What is the advantage of very small particle sizes in LSC columns? (b) With reference to the Va...
A: a) Small particle sizes are used in the LSC columns.
Q: Given the reaction: CuSO4.3H2O CuSO4.2H2O + H2O the DELTA H° = 9900 and DELTA G°=4500 at 25°C. Cal...
A: The given reaction is : CuSO4·3H2O(s) ↔ CuSO4.2H2O(s) + H2O(g) ∆H° = 9900 J/mol and ∆G°=4500 J/m...

Step by step
Solved in 3 steps with 3 images

- OneLogin ALEKS - Kayley Valentine b nickel (II) sulfate cation and ani x Lea x + A www-awu.aleks.com/alekscgi/x/Isl.exe/1o_u-IgNslkr7j8P3jH-IBI5ahvJxLWwcNXdqGDInlqlcnbb-P21X0OTA0ITA3YJ_7HPrxSXe3aRFsU.. Apps O R Prechecks: Test 2... O ATOMS, IONS AND MOLECULES Deducing the ions in a polyatomic ionic compound from its empir... Complete the table below by writing the symbols for the cation and anion that make up each ionic compound. The first row has been completed for you. ionic compound cation anion NaCl CI Na NiSO, V Cl, Cro, Fe (OH), Explanation Check O 2020 McGraw-Hill Education. All Rights Reserved. Terms of Use l Pr MaćBook Pro esc 888 F4 F1 F5 F6 F7 F8 F9 F10 F11 F12 24 & Q т H. alt olt tion command command optic * 00Q2: (A) Sketch the following Miller indices: 1. [ī02] 2.(ziio) . (312)C. Cr(C104)3 Show Hint
- The value of 10Dq in the complex K3[Co(C204)3] is ~=-m~=-- the * complex [Co(H20)6]CI3 less than in O bigger than in O equal to O The complex [Co(H20)6]CI2 is less stable than the complex * [Zn(H20)6]CI2 Error |:| true |:| The Na2[CoBr4] complex is more * stable than the K2[CoCl4] complex Error |:| true |:|STANDARDIZE THE Na,S:Os.SH2O WITH KIO.. Weight of KIO, = 0.5325 g Vot KIO: = 250 ml N KIO, V KIOL ml. 7.0 7.0 VNas:O1.5H0, mL 8.2 7.5 N Nas2O.5H;0 Average N Nays:0,5HOGiven the following size frequency for a dust: Size Interval (um) 7-17.5 17.5-21 21-25 25-28 28-30 30-33 33-36 36-41 41-49 49-70 % by Number 10 10 10 10 10 10 10 10 10 10 a. Plot the cumulative frequency distributions (in %) of the number, surface area, and mass on linear graph paper assuming all particles are spheres with Pp 1.6 g cm ³. = b. Is this a log-normally distributed dust?
- Chlorine pentafluoride D (156) TESK - Green Sta B Final assessmentpdf - T Lab Reports - 202030 A www-awu.aleks.com/alekscgi/x/lIsl.exe/1o_u-IgNslkr7j8P3jH-lv-OZhvG1qYPWviNXOF96XoVW3_W7J0tF-2drAGbNSolpTW6rAkbtcy5PbEwUBck_5y4DIn88jVEEPfKWHKeGb8Pd1BB-lo?1oBw Gmail YouTube A Home - SUNY Broo. SUNY Broome Com. = Calculus Preface (?) E Brytwave Blackboard O Clutch Quizlet Knowledge Check Question 9 A student dissolves 14.9 g of potassium chloride (KCI) in 200. g of water in a well-insulated open cup. He then observes the temperature of the water fall from 20.0 °C to 15.2 °C over the course of 8.9 minutes. Use this data, and any information you need from the ALEKS Data resource, to answer the questions below about this reaction: KCl(s) → K (aq) + Cl (aq) You can make any reasonable assumptions about the physical properties of the solution. Be sure answers you calculate using measured data are rounded to the correct number of significant digits. Note for advanced students: it's possible the…The relative intensities for caffeine are m/z 195/194 5 10.3%. What is the isotopic composition of the ion at m/z 195? What is the expected intensity ratio m/z 195/194?Chrome File Edit View History Bookmarks Profiles email draft - Google Docs sc ● Consider this reaction: 1 A ALEKS- Lara Althawadi - Lear X ☆ C www-awu.aleks.com/alekscgi/x/lsl.exe/1o_u-IgNsikr7j8P3jH-IvTqeviKFP6W0cqJcWJdIACROQwyw24GWHInUUz9jGCwQIcISUzhOQhHxz_5bM9yeRdQ... 17.4 Solubility and.... 5.3 Enthalpies of... 18.5 Gibbs Free E...Reading Schedule 19.6 Reduction Po... Y SOLUTION: The le... Math 115 W-S Fall... 2H, PO₂ (aq) → P₂O, (aq) + 3H₂O (aq) - At a certain temperature it obeys this rate law. -1 rate = (82. M¹-s¹) [H₂PO4] Q A 18.3 Gibbs Free E... O KINETICS AND EQUILIBRIUM Using first- and second-order integrated rate laws Os Explanation F N 18.210 @ x Nancy Foldi - Department of PX Suppose a vessel contains H₂PO4 at a concentration of 0.290M. Calculate how long it takes for the concentration of H₂PO4 to decrease by 92.%. You may assume no other reaction is important. Round your answer to 2 significant digits. 2 W Check S # X :m 3 26 9.f E Tab D 14 $ 4 Window Help C X R Ś F JO…
- Calculate e° values for the galvanic cells based on the following overall reactions. Assume that all concentrations are 1.0 M and that all partial pressures are 1.0 atm.what is the thore tical yield of vanadium that can be produced y the reaction of 40.09 of V 205 with 40.0s of calcium based on the followin) chemiAl reacton V 205 CS) +5 Cal ) 2Va) +S CaD cS) 6-) 1.23 bi) 22ug c)stg d.) 40.05 e) 20:35Determine the isotopic peak scheme for [ReCl]+ fragment in the mass spectrum? 185Re: %37 ; 187Re: %63; 35Cl: %76; 37Cl: %24

