
Concept explainers
(a)
Interpretation: The Newman projections of
Concept introduction: Various interconvertible forms that result from rotation around the
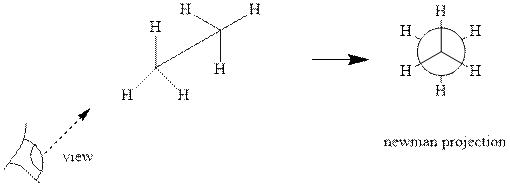
As rotation is carried out along

(b)
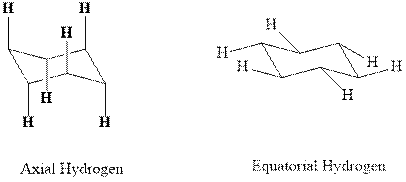
Interpretation: The indicated molecules should be drawn in chair conformations.
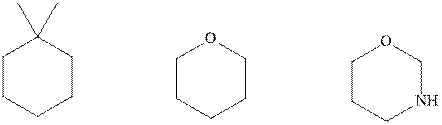
Concept introduction:In chair form, all the
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
Organic Chemistry: Structure and Function
- 4. (a) Draw a skeletal (line-bond) structure for 3,4-dimethylhexane. (b) Draw a sawhorse representation of any staggered conformation of this molecule looking down the carbon-3 to carbon-4 bond. (c) Draw a Newman projection looking down the carbon-3 to carbon-4 bond of the same conformation that you drew as a sawhorse representation.arrow_forwardDraw the structures of 1,3-pentadiene and 1,4-pentadiene and label the carbons. Predict the trendin C-C single bond lengths in the two compounds.arrow_forwardDraw a dash-wedge structure for (1R)-1-bromo-1,3,3-trimethylcyclohexane.arrow_forward
- Following is the Structural formal of Chloropropane H H H 2. H Draw Newman projections for all staggered and eclipsed conformation formed by rotation about C1 - C2 bond from 0° to 360°. b) Which Staggered conformation(s) has the lowest energy; which has the highest energy? c) Which eclipsed conformation(s) has the lowest energy, which has thhe highest energy?arrow_forwardConsider 1-bromopropane, CH3CH2CH2Br. (a) Draw a Newman projection for the conformation in which CH3 and -Br are anti (dihedral angle 180°). (b) Draw Newman projections for the conformations in which - CH3 and -Br are gauche (dihedral angles 60° and 300°). (c) Which of these is the lowest energy conformation? (d) Which of these conformations, if any, are related by reflection?arrow_forward-1. What is the equivalent bond-line formula for the molecule, HOCH-CH(CH-CH₂)-CH(CH)? HO. (B) HO (C) HO. -2. What is the equivalent structure for this molecule using a hond-line formula? A) C) Br Br (D) (B) HO (CH₂)2CH(CH₂) CHBICHCH, Br CH₂CH3 CH3 yazparrow_forward
- (5) Draw the most stable chair conformation of the cyclohexane shown below. CH3 CI H3C Br OH CH3arrow_forwardSighting along the appropriate C-C bond of neopentane C(CH3)4: a) Draw its Newman projections. b) How much energy does the most unstable conformation possess? c) Make the energy diagram for the different conformers.arrow_forwardQ2. Which one of the following structures is the most stable conformation for 2-bromo butane (viewing down the C2-C3 bond axis where C2 is front carbon and C3 is back carbon) Br H H H **** CH, CH₂ (B) Br (A) H H (D)arrow_forward
- 6. Newman projections to Lewis structures From the following Newman projections, draw the corresponding Lewis structure in the same conformation. A) B) H. H CH3 H₂C N H OH CI Br CH3 H OH CH3arrow_forward13a. Draw the correct skeletal structure of 5-methyl-1-hexanol. provided (13b.) draw in skeletal structure its most stable conformer looking along C3-C4 (* hydrogens that are directly connected to the Newman projection ( and uii the Newman Projection ;; Draw the 13a. 13b. draw the correct skeletal structure here draw the correct skeletal structure here 4arrow_forward• -- H (b) H,C 3-methylhexane CH, (c) B கூ CH, CH, HgCG; சீகூ H₂CH H 3-ethyl-1-methyloctane 3,3 diethyl pentane 5Å, Å,CH, W CH3 CH2 CH₂ W21 Su2... CH3 X -CH3 G b F E L carrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





