
Concept explainers
1. Use the following graph to diagram the energetics of a
To describe:
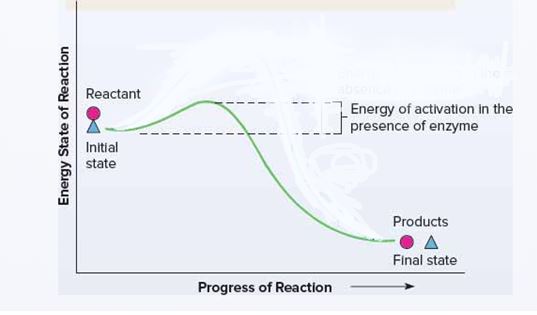
The energetics of a chemical reaction with and without an enzyme. To position reactants and products at appropriate points and to indicate the stages in the reaction and the energy levels.
Introduction:
Enzymes are remarkable example of protein biocatalyst that facilitates metabolic reactions.
Explanation of Solution
Enzymes increase the rate of a chemical reaction without becoming part of the products or being consumed in the reaction. Because of the free energy inherent in molecules, a reaction could occur spontaneously at some point even without an enzyme but at a very slow rate.
Energitics of a chemical reaction with an enzyme:
Energitics of a chemical reaction without an enzyme:
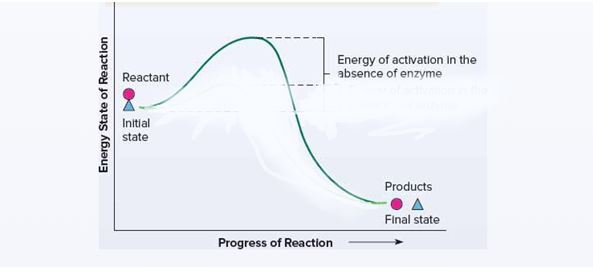
Enzymes increase the rate of a chemical reaction without becoming part of the products or being consumed in the reaction.
Want to see more full solutions like this?
Chapter 8 Solutions
Foundations in Microbiology
- 2. Draw a graph showing a downhill (spontaneous) reaction, that requires some activation energy to get it going. Then draw and clearly label on the graph b. The activation energy for the reaction (use a bracket to indicate the upper and lower limits) c. The free energy change for the reactions (use a bracket to indicate the upper and lower limits) d. How an enzyme will change the energetics of the reaction. I suggest you use a dotted line for the enzyme.arrow_forward8). Which statement best describes 1 point the enzyme represented in the graphs? O 10 20 30 40 50 60 O 2 4 6 10 12 Temperature ("C) PH (1) This enzyme works best at a temperature of 50°C and a pH of 12. (2) Temperature and pH have no effect on the action of this enzyme. (3) This enzyme works best at a temperature above 50°C and a pH above 12. (4) This enzyme works best at a temperature of 35°C and a pH of 8. Relative Rate of Enzyme Action Relative Rate of Enzyme Actionarrow_forward1. Match the following words with their definitions. Product Active site Enzymes Catalyst Substrate Activation energy a. amount of energy required for a chemical reaction to occur b. substances that bring about a chemical reaction without being changed itself c. substances that enzymes act upon d. regions on the surface of enzymes that fit the substrate e. substance formed from the substrate at the end of the chemical reaction with an enzyme f. proteins that speed up a chemical reactionarrow_forward
- 1. a. What is the abbreviated name for the molecule below? (3 letters) What is the abbreviated name of this molecule if it has two phosphates? What is the abbreviated name of this molecule if it has one phosphate? From your answers above, circle the form of the molecule that has the most energy. What type of energy is this? circle all that apply: (KINETIC / POTENTIAL / CHEMICAL) Where is the energy? b. Identify the three main parts of this molecule. (write on the red brackets i-iii) c. Parts ii and ii together make (blue vertical bracket) ii. NH2 НО-Р ОН ОН ОН ОН ОН iii d. What is the specific name for this molecule? e. This molecule is also a monomer building block of which biomolecule (be specific) The general name for this type of monomer is 2. a. Put arrows by the two high energy bonds on the molecule above. Explain why these functional groups are difficult to join. (hint: these are acids. Circle the H that will be donated at cellular pH). (MORE / LESS/ the SAME amount of) energy…arrow_forward4. Using a fluorescent model substrate, you study the kinetics of an enzyme-catalyzed reaction. You observe the below data. Write an equation describing reaction velocity (v) versus [S]. Define and provide a numerical value for any constants that you include in your equation. 16 14 12 4 2 0.0 0.5 1.0 1.5 2.0 2.5 3.0 3.5 [S] / mM `N' at 50 mM and observe the following In pilot experiments, you add the putative inhibitor imidazole Doesn't affect anything 1 reaction velocities: [S] = 1 mM5 µM min-1 [S] = 2 mM9 9 µM min-1 [S] = 4 mM 911 µM min-1 Is imidazole a competitive or non-competitive inhibitor? Provide a brief explanation. > 9 00 v/ µM min-arrow_forward4. Consider the reaction: OH CH H3C O C H₂ a. What kind of reaction is being performed here? H3C C H₂ b. What enzyme performs this reaction? c. What cofactors, if any, are required for this reaction?arrow_forward
- 7. Which of the following statements is true about enzyme-catalyzed reactions? The reaction is faster than the same reaction in the absence of the enzyme b. The free energy change of the reaction is opposite from the reaction without the enzyme The reaction always goes in the direction toward chemical equilibrium d. Enzyme-catalyzed reactions require energy to activate the enzyme e. Enzyme-catalyzed reactions release more free energy than noncatalyzed reactions The following questions are based on the reaction A+B C+D Free Energy- A+B Progress of the Reaction C.Darrow_forward1. Below is a plot that shows the relative activity of a particular enzyme at various Answer the following questions about this graph. temperatures. Enzyme activity → 10 15 20 25 30 35 40 45 50 Temperature (°C). A. Why does the enzyme's activity increase between 10°C and 37°C? ( B. Why does the enzyme's activity decrease sharply after the temperature is raised above 37°C?(arrow_forwardIn a transition state diagram, which of the following are features of the transition state (TS)? There may be more than one correct answer, select all that apply. The change in energy in ground state to the transition state represents the Gibbs Free Energy If the reaction is reversible, the TS will only progress forward to form products The TS occupies a trough The TS is associated with the highest energy The TS occupies the highest peakarrow_forward
- 8. What enzyme breaks peptide bonds which occur to the carboxyl terminal side of aromatic amino acids? Relate the pH profile of this enzyme to its catalytic mechanism. HINT: Draw out the pH profile and initial steps of the mechanism for this enzyme.arrow_forward1 The Asp residue with a pka = 6 is found in the active site of an enzyme. This residue acts as an acid catalyst. Assuming that the enzyme is stable at a wide range of PH and that the Aspartic acid residue is the only ionizable residue in the active site, please draw the curve of enzyme activity versus pH on the graph below and explain the shape of your curve. (10 points) 100 % enzyme activity 75 50 25 3 LO 5 7 pH T 9 11arrow_forwardDescribe how enzymes speed up chemical reactions (both energetically and physically!), and how they affect the energy and equilibrium of a reaction. Describe 6 different physical and chemical factors that can regulate enzyme activity.arrow_forward
 Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Biology: The Dynamic Science (MindTap Course List)BiologyISBN:9781305389892Author:Peter J. Russell, Paul E. Hertz, Beverly McMillanPublisher:Cengage Learning
Biology: The Dynamic Science (MindTap Course List)BiologyISBN:9781305389892Author:Peter J. Russell, Paul E. Hertz, Beverly McMillanPublisher:Cengage Learning

