Sodium carbonate is a reagent that may be used to standardize acids in the same way that you have used KHP in this experiment. In such a standardization it was found that a 0.498 -g sample of sodium carbonate required 21.7mLmL of a sulfuric acid solution to reach the end point for the reaction. Na, CO, (aq) + H₂SO (aq) - H₂O(l) + CO₂ (g) + Na SO (ag) What is the molarity of the H, SO? 2 3 2 4 2 2 2 2 4 Sodium carbonate is a reagent that may be used to standardize acids in the same way that you have used KHP in this experiment. In such a standardization it was found that a 0.498-g sample of sodium carbonate required 21.7 mL mL of a sulfuric acid solution to reach the end point for the reaction. Na2CO3(aq) + H2SO4(aq) + H2O(l) + CO2 (g) + Na2SO4(aq) What is the molarity of the H2SO4? ΜΕ ΑΣΦ Submit Request Answer ? M
Sodium carbonate is a reagent that may be used to standardize acids in the same way that you have used KHP in this experiment. In such a standardization it was found that a 0.498 -g sample of sodium carbonate required 21.7mLmL of a sulfuric acid solution to reach the end point for the reaction. Na, CO, (aq) + H₂SO (aq) - H₂O(l) + CO₂ (g) + Na SO (ag) What is the molarity of the H, SO? 2 3 2 4 2 2 2 2 4 Sodium carbonate is a reagent that may be used to standardize acids in the same way that you have used KHP in this experiment. In such a standardization it was found that a 0.498-g sample of sodium carbonate required 21.7 mL mL of a sulfuric acid solution to reach the end point for the reaction. Na2CO3(aq) + H2SO4(aq) + H2O(l) + CO2 (g) + Na2SO4(aq) What is the molarity of the H2SO4? ΜΕ ΑΣΦ Submit Request Answer ? M
Related questions
Question
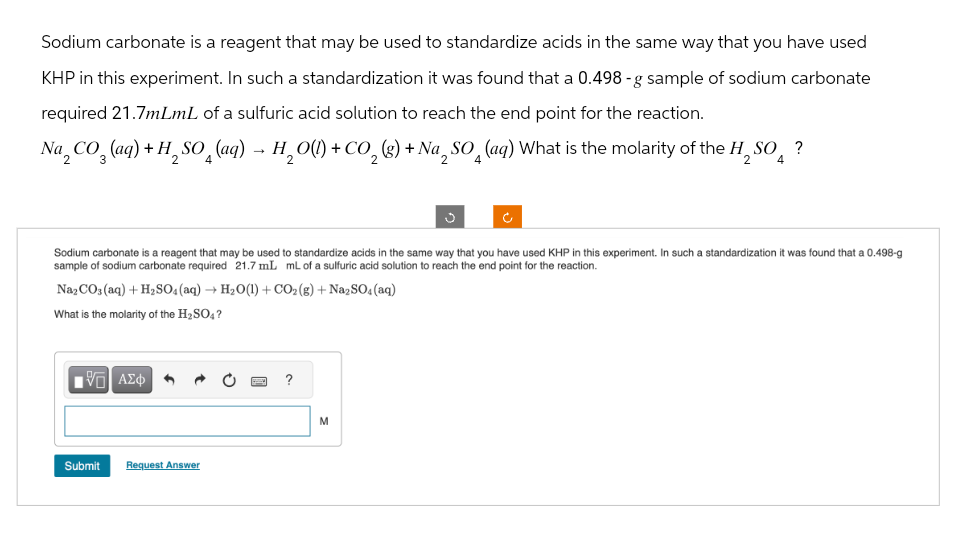
Transcribed Image Text:Sodium carbonate is a reagent that may be used to standardize acids in the same way that you have used
KHP in this experiment. In such a standardization it was found that a 0.498 -g sample of sodium carbonate
required 21.7mLmL of a sulfuric acid solution to reach the end point for the reaction.
Na, CO, (aq) + H₂SO (aq) - H₂O(l) + CO₂ (g) + Na SO (ag) What is the molarity of the H, SO?
2
3
2
4
2
2
2
2 4
Sodium carbonate is a reagent that may be used to standardize acids in the same way that you have used KHP in this experiment. In such a standardization it was found that a 0.498-g
sample of sodium carbonate required 21.7 mL mL of a sulfuric acid solution to reach the end point for the reaction.
Na2CO3(aq) + H2SO4(aq) + H2O(l) + CO2 (g) + Na2SO4(aq)
What is the molarity of the H2SO4?
ΜΕ ΑΣΦ
Submit
Request Answer
?
M
AI-Generated Solution
Unlock instant AI solutions
Tap the button
to generate a solution