Select the chromate compound that is most soluble in water at 25°C on a molar basis using the following solubility data. Ksp BaCrO4 2.0 x 10-9 PbCrO4 1.8 x 10-14 Ag2CrO4 7.0 x 10-12
Select the chromate compound that is most soluble in water at 25°C on a molar basis using the following solubility data. Ksp BaCrO4 2.0 x 10-9 PbCrO4 1.8 x 10-14 Ag2CrO4 7.0 x 10-12
Chapter6: The Systematic Approach To Equilibria: Solving Many Equations
Section: Chapter Questions
Problem 10P
Related questions
Question
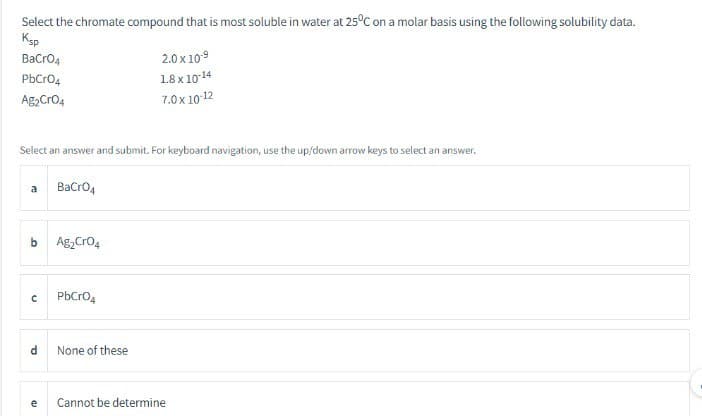
Transcribed Image Text:Select the chromate compound that is most soluble in water at 25°C on a molar basis using the following solubility data.
Ksp
BaCrO4
PbCrO4
Ag2CrO4
2.0 x 10-9
1.8 x 10-14
7.0 x 10-12
Select an answer and submit. For keyboard navigation, use the up/down arrow keys to select an answer.
a BaCrO4
b Ag₂CrO4
с PbCrO4
d
a
None of these
Cannot be determine
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax



Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT