Q: The enthalpy change for the following reaction is given below. Pb (s) + CO2 (g) → PbO (s) + CO (g)…
A: We have to use the formula- ∆Horex=∑n∆Hoproduct-∑n∆Horeactant for the reaction Pb(s) + CO2(g) → PbO…
Q: Why is it essential to practice on how to prepare solutions (stock solution/dilutions)? How does…
A: The preparation of solutions is a fundamental concept that is essential in many scientific and…
Q: Which compounds are involved in double replacement reactions? O ionic compounds O polar covalent…
A: we have to determine the compounds that give double replacement reaction
Q: 5. Provide a mechanism and product for the reaction below using formal arrow pushing. Show how the…
A:
Q: How much energy is required to melt 882.2 g of methanol at its melting point?
A: The ∆Hfusion of methanol = 3.22 kJ/mol The heat required to melt methanol = Q = N x ∆H Molar mass of…
Q: Indicate the FALSE statement regarding average and instantaneous rates of reactions: Question 9…
A: The above question is asking for the false statement among four options regarding average and…
Q: How many grams of PCl5 contain 2.55 x 10^25 atoms of Cl in it? Show work.
A:
Q: Suppose i want to prepare/ manufacture a 20 litter of a shampoo used in car or bike wash, what are…
A: Solutions- Preparation method of shampoo.
Q: Chloroform, CHCl3 is a volatile liquid and has a boiling point of 61.7 ° C and has a heat of…
A:
Q: What is Molar heat of vaporization? ANSWER: For this question, write the formula and write the…
A: Vaporization is the process by which a substance changes from its liquid state to its gaseous state…
Q: The treatment of PhBr with n-BuLi yields: ות
A: The free radical mechanism is a type of chemical reaction in which a molecule reacts by removing an…
Q: Cyclohexene has the formula C6H₁0 and the structure shown in Figure 4-4. When cyclohexene is treated…
A: Given reactant are cyclohexene. Alkene reacts with acid and water undergoes hydration reaction and…
Q: A certain reaction is known to have an activation energy E,-8.0 kJ/mol. Furthermore, the rate of…
A: Answer: These question are based on Arrhenius equation that gives us relation between rate of…
Q: Calculate AG for the following reaction at 25 °C. C3Hg (g) +50₂(g) - 1 3CO₂(g) + 4H₂0 (1) Be sure…
A:
Q: What are the resonances?
A: Given : structure of molecule
Q: A solution is made using 10.5 percent by mass CH₂Cl₂ in CHCl₃. At 30 °C, the vapor pressure of pure…
A: •Here we have to calculate boiling point of solution. => Given:- 1) Boiling point of pure CHCl3 =…
Q: 2. Using Excel, make a graph of the following data. Plot Mass vs. Volume for the following data on…
A: The given data of mass of Al and mass of Zr and volume is: Volume (mL) Mass of Al (g) Mass of Zr…
Q: Question 1 Consider the reaction: 2 A (aq) ⇌ B(aq) Given the following KC values and starting with…
A: The given equilibrium equation is; 2 A (aq) ⇌ B(aq) The equilibrium constant for the given equation…
Q: Why do ionic and polar covalent substances conduct electricity and nonpolar molecules are not…
A: Ionic and polar covalent substances conduct electricity because they contain ions or polar molecules…
Q: At constant volume, the heat of combustion of a particular compound, compound A, is −3046.0…
A: According to the question, The mass of compound A = 1.753 gThe molar mass of the compound A = 112.07…
Q: See Figure 4-6. Which structure is an incorrect resonance form for the benzylic cation? A B U
A: Any carbocation in which the positive formal charge and open octet are situated adjacent to a…
Q: P3.6 A vessel is filled completely with liquid water and sealed at 13.56°C and a pressure of 1.00…
A: Given , Temperature , T = 13.56 °C=(13.56+273)K=286.56 K Initial pressure , Pi=1 bar
Q: Provide the correct common name for the compound shown here. H₂C CH3 tri tert- CH3 di cyclo iso sec-
A: Introduction The International Union of Pure and Applied Chemistry (IUPAC) has established a system…
Q: See Figure 4-5. Match the spectrum to its likely structure. A B C D [Choose ] [Choose ] [Choose ]…
A: IR spectroscopy is mainly used for the identification of functional group present in the molecule.…
Q: 17. If you dissolve 20.0 grams of glucose (C6H1206) in 500.0 mL of water, what will its osmotic…
A:
Q: Draw the products of the following reaction: CH₂CH=CHCH₂- + H₂O H₂SO ? (two possible products) Draw…
A: Addition of water molecule across C=C bond to form alcohol according to markovnikov's rule.Rule…
Q: 7. Convert the following (Show your work) () a. 75.14 µg/ml →ng/ul b. 743.2 µmol → nmol c. 55 ng →…
A:
Q: Which set of quantum numbers cannot be correct? A. n = 1, l = 1, ml = 1 B. n = 2, l…
A: for principal quantum number n, azimuthal quantum number (l) values can be integers from 0 to n-1.…
Q: 5. Which of the compounds below would have a lower melting point? Circle your answer and explain why…
A: “Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: 1) Describe at least three systematic errors that might occur while weighing a solid on an…
A: Please find detailed answer of both :-
Q: k₁[A] [B] k2k3 [A] [D] 2k1k3[A]²[B] k2[D] 2k₁[A][B] k₁[A] [B] k3 [A] k₁[A][B]-k₂[C][D]-k3[A][C]…
A:
Q: you have a 0.05M stock solution of NaCl, a 0.25M stock solution of glucose, and a bottle of solid…
A: Given: Concentration of stock solution of NaCl is 0.25M Concentration of stock solution of glucose…
Q: Ar] 4s2 3d10 4p3 is the electron configuration of: Group of answer choices Sb V As P
A: Total Number of electrons in ground state electronic configuration= Atomic number of element The…
Q: Water has a vapor pressure of 18.0 torr and a density of 0.997 g/mL. Acetone (CH3COCH3) has a vapor…
A:
Q: CEA EXCEA a Ama W CEA…
A: We know Kp of forward reaction with half number of moles is the square root of Kp of forward…
Q: 2. Identify the following molecules as chiral or achiral. e) f) Br O 09 OH { OH h) OH
A: Given : structure of molecules Tip : check POS and COS
Q: Suppose you are assigned to manufacture or prepare a 20 litter shampoo used in bike or car washing.…
A: The scientific mechanism of action of shampoo involves the surfactants present in the shampoo.…
Q: Given the equation: −2A(g) + B(s) = 3C(g) + D (g) Which of the following actions will cause the…
A: Given the equation: 2A(g) + B(s) <=> 3C(g)+D(g) Which of the following actions will cause the…
Q: Examine the starting FeCl and FeCI, solids used to prepare magnetite (leave the solids in their…
A: Magnetite (Fe3O4) is a naturally occurring iron oxide mineral. It is black or brownish-black in…
Q: How many of the following three choices have hydrogen bonding as their strongest intermolecular…
A: We need to find how many of the following three choices have hydrogen bonding as their strongest…
Q: In a beaker at 25 C and 1 atm pressure, 100 g of zinc are used to react with dilute H2SO4. What is…
A: Given that At 25 °C and 1 atm pressure, 100 g of zinc are used to react with dilute H2SO4. The work…
Q: Standard Enthalpy of Formation and Hess's Law 15. Consider the following data: ΔΗ° (kJ) -62.8 -635.5…
A: Hess's law describes that regardless of the multiple stages or steps of a reaction, the total…
Q: Two gases were allowed to react in a chamber. SO2 gas (0.2 M) and O2 gas (0.2 M) were reacted at…
A:
Q: How much does 1.29x10^25 particles of HCl weigh? Round to 2 decimal places.
A: In this question, we will determine the weight of the given particle HCl in the given number of…
Q: Place the following three choices in order of increasing melting point. Xe Kr Ar
A:
Q: Arrange the following compounds in order of elution from a column of silica gel: A) 1,3-…
A: Explanation The order of elution in column chromatography with silica used as column is determined…
Q: With the aid of the following balanced equation and molar masses (MM), determine the answer for the…
A: Given - Molar mass of C6H10= 82.2 g/mol Molar mass of O2= 32 g/mol Molar mass of CO2= 44g/mol Molar…
Q: Describ the physical properties of Co2 and,also draw a phase diagram of Co2
A: Phase diagram is basically a graph of physical state of any pure substance existing at different…
Q: ow much energy is required to boil 551.8 g of methanol at its boiling point of 64.7 °C?
A: The energy required to boil a substance, also known as the enthalpy of vaporization, can be…
Q: 15. What is the freezing point for an aqueous solution of KBr that was made from dissolving 25.0…
A:
How many grams of Cl2O7 contains 2.85 x 1021 molecules of Cl2O7 in it? Show work.
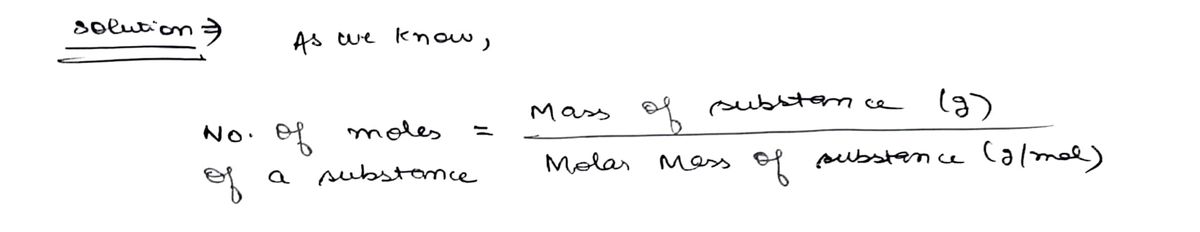
Step by step
Solved in 2 steps with 2 images

- What is the mass, in grams, of 0.0331 mole magnesium iodide? SHOW WORK.Calculate the number of moles in each sample show work? a. 72.5g CCI4 b. 12.4g C12H22O11 c. 25.2 kg C2H2 d. 12.3 g dinitrogen monoxidehow many grams of hydrogen are contained in 2.50 x 10^23 molecules of ammonium phosphate show work
- Abdelra X M Mathway | Algebra Prol X G molar mass of zinc chl x Launch Meeting-Zoom x G 0.16kg to g-Google Se x+ www-awu.aleks.com/alekscgi/x/Isl.exe/1o_u-IgNslkr7j8P3jH-lix5uFZIVj2iEJjQd1WoxT77ErtzZpGbzESWMk5SbMzyGfsmkwQSq2n2qsprma6JcCeltVTwZDkOLh51Rytx20V. O GASES, LIQUIDS, AND SOLIDS Using the Kf and Kb equations with electrolytes Jeneen v A certain liquid X has a normal boiling point of 93.40°C and a boiling point elevation constant K=1.46 °C-kg mol . A solution is prepared by dissolving -1 9. some iron(III) nitrate (Fe(NO,)) in 550. g of X. This solution boils at 93.8 °C. Calculate the mass of Fe(NO,) that was dissolved. Be sure your answer is rounded to the correct number of significiant diglts. x10 Check Explanation 2021 McGraw-Hill Education. All Rights Reserved. Terms of Use Privacy Accessibility M9E DetaMath 6 Quiz Chemistry Spring Final x D A sample of helium gas has xG 391ml of gas with a tempera x G number of electrons on a tric + -> b cfbisd.instructure.com/courses/85698/quizzes/390093/take/questions/2865049 Q * e D GO D CFB A Pearson Realize 6 Dashboard @ Student Resources E Google Docs creat A google ciassroom O Nearpod Welcome- O Meeting is in progre O Reading list Balance the following reaction: Al + Fe203 → Al203 + Fe *remember to enter a 1 for coefficient is there is no change. US VO 3:53 esc @ #3 $ bac 3 4 7 8. 9. W e t y a d. f k80 grams of Potassium (K) was put in 3000 ml of water. They underwent a violent reaction and produced hydrogen gas which was allowed to mix with 20 g of air. Find the average molecular weight of that mixed gas stream. [Ref: 16O, 14N, 39K, 1H]
- Previous Page 6 of 16 Next O References Use the References to access important values if need According to the following reaction, how many grams of oxygen gas are required for the complete reaction of 25.2 grams of ammonia? ammonia (g) + oxygen (g) nitrogen monoxide (g) + water (g) grams oxygen gas Cengage Learning | Cengage Technical Supportock MISSED THIS? Read Section 6.6 (Pages 228-235); Watch KCV 6.6, IWE 6.10 A gas mixture contains 1.29 g N₂ and 0.76 g O₂ in a 1.64-L container at 15 °C. esc Mother to Son &....pdf ! 1 ĐỘ F1 Q A N @ 2 * 38 F2 W S #3 80 F3 E D $ 4 X C Q F4 Part C Calculate the partial pressure of N₂. Express your answer using two significant figures. P₁ = Submit Part D R P₂ = Calculate the partial pressure of O2. Express your answer using two significant figures. Submit F [ΨΕΙ ΑΣΦ % 5 Request Answer [5] ΑΣΦ Request Answer ļ F5 T V 1 6 MacBook Air G → F6 & 7 H F7 ? - ? BN atm atm * 8 31 DII F8 ( 9 F K M1. Hydrogen peroxide is decomposed in a lab and the produced O2 (g) is collected over water at 15.0 °C. Molar masses: H2O2 = 34.02 g/mol, H2O = 18.02 g/mol, O2 = 32.00 g/mol. 2H2O2 (l) --> 2H2O (l) + O2(g) a) Calculate the moles of O2 (g) collected if the total pressure of the gas is 780. mmHg and the volume of gas is 0.4000 L. Assume all gases are ideal. b) What volume of pure hydrogen peroxide (density = 1.530 g/mL) decomposed? Assume that the decomposition reaction is complete. If you were unable to answer part (a), assume that 1 mol of O2 (g) was collected. c) If the collected gas (H2O (g) + O2 (g)) is transferred into a rubber balloon (which contains tiny pores), which gas will effuse out more quickly? Briefly explain (in about 1 sentence) why your answer makes physical sense. You can ignore the difference in partial pressures for this problem.
- 17. A fuel mixture used in the early days of rocketry consisted of two liquids, hydrazine (N2H4) and dinitrogen tetraoxide (N2O4), which ignite on contact to form nitrogen gas and water vapor. How much in grams of the excess reagent is used up when 435.0 g of N2H4 and 718.5 g of N2O4 are mixed? (, MW: N204 = 92.011g/mol, N2H4 = 32.05g/mol, N2 =28.013 g/mol 2N2H4(0) + N2O40) –> 3N2(9) + 4H2O(g)Determine the Kinetic energy of 1 mole oxygen at 250 K. (R = 8.314 J/K.mol). (20 pts)X 1. DME, heat 2. LIAIH4, THF 3. dilute HCI, H₂O i 4. Cl3C H₂SO4 (cat.)

