Consider the equilibrium system described by the chemical reaction below, which has a value of Kc equal to 7.1 x 10^-4 at a certain temperature. If a solid sample of CaCrO4 dissolves in a solution, what will the equilibrium concentration of Ca2+ in the solution be? 1. Based on the given values, set up ICE Table in order to determine the unknown 2. Based on your ICE table, set up the expression for Kc in order to determine the unknown. Do not combine or simplify terms. 3. Based on your ICE table and expression for Kc, solve for the concentration of Ca2+ at equilibrium.
Consider the equilibrium system described by the chemical reaction below, which has a value of Kc equal to 7.1 x 10^-4 at a certain temperature. If a solid sample of CaCrO4 dissolves in a solution, what will the equilibrium concentration of Ca2+ in the solution be? 1. Based on the given values, set up ICE Table in order to determine the unknown 2. Based on your ICE table, set up the expression for Kc in order to determine the unknown. Do not combine or simplify terms. 3. Based on your ICE table and expression for Kc, solve for the concentration of Ca2+ at equilibrium.
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter12: Chemical Equilibrium
Section: Chapter Questions
Problem 12.41PAE: Because calcium carbonate is a sink for CO32- in a lake, the student in Exercise 12.39 decides to go...
Related questions
Question
Consider the equilibrium system described by the
1. Based on the given values, set up ICE Table in order to determine the unknown
2. Based on your ICE table, set up the expression for Kc in order to determine the unknown. Do not combine or simplify terms.
3. Based on your ICE table and expression for Kc, solve for the concentration of Ca2+ at equilibrium.

Transcribed Image Text:Question 6 of 18
Consider the equilibrium system described by the chemical reaction
below, which has a value of Kc equal to 7.1 × 10-4 at a certain
temperature. If a solid sample of CaCro, dissolves in solution, what will
the equilibrium concentration of Ca2+ in the solution be?
CaCrO:(s) = Ca²"(aq) + CrO,2-(aq)
1
2
3
NEXT >
Based on the given values, set up ICE table in order to determine the unknown.
СаcrO-(s)
Ca2-(aq)
Cro.2 (aq)
Initial (M)
Change (M)
Equilibrium (M)
5 RESET
7.1 x 10-4
+x
-x
+2x
-2x
7.1 x 104 + x
![Question 6 of 18
Consider the equilibrium system described by the chemical reaction
below, which has a value of Kc equal to 7.1 × 10-4 at a certain
temperature. If a solid sample of CaCros dissolves in solution, what will
the equilibrium concentration of Ca2- in the solution be?
CaCrO:(s) :
— Са" (аq) + CrO.2 (aq)
PREV
1
2
NEXT
Based on your ICE table, set up the expression for Kc in order to determine the
unknown. Do not combine or simplify terms.
Ko
= 7.1 x 10-4
RESET
[x]
[2x]
[2xP
7.1 x 104 + x]
[7.1 x 10-4 - x]
[7.1 x 104 + 2x]
[7.1 x 104 - 2x]
[7.1 x 104 + xr
[7.1 x 104 - x]?
[7.1 x 104 + 2x
[7.1 x 104 - 2xP](/v2/_next/image?url=https%3A%2F%2Fcontent.bartleby.com%2Fqna-images%2Fquestion%2F974c41b3-3730-44f8-a8cd-a433355f091c%2Fa14f0e18-f6ce-4b30-8e6e-ad3afb75c6b3%2Fzk688rg_processed.png&w=3840&q=75)
Transcribed Image Text:Question 6 of 18
Consider the equilibrium system described by the chemical reaction
below, which has a value of Kc equal to 7.1 × 10-4 at a certain
temperature. If a solid sample of CaCros dissolves in solution, what will
the equilibrium concentration of Ca2- in the solution be?
CaCrO:(s) :
— Са" (аq) + CrO.2 (aq)
PREV
1
2
NEXT
Based on your ICE table, set up the expression for Kc in order to determine the
unknown. Do not combine or simplify terms.
Ko
= 7.1 x 10-4
RESET
[x]
[2x]
[2xP
7.1 x 104 + x]
[7.1 x 10-4 - x]
[7.1 x 104 + 2x]
[7.1 x 104 - 2x]
[7.1 x 104 + xr
[7.1 x 104 - x]?
[7.1 x 104 + 2x
[7.1 x 104 - 2xP
Expert Solution
Step 1
The given reaction is :
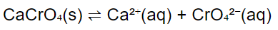
Kc for the reaction = 7.1 x 10-4
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning