Complete the balanced dissociation equation for the compound below in aqueous solution. If the compound does not dissociate, write NR after the reaction arrow. NiS(s)-->
Q: Calculate the number of moles of: sodium hydroxide in 100 cm³ of 0.1 mol dm³ sodium hydroxide soluti...
A: The number of moles = Molarity×volume×10-3
Q: Calculate the mass of 1.0 mmol nitro ester [2-(diethylamino)ethyl 4-nitrobenzoate] Mw: 266.3 g/mol
A:
Q: Label each molecule with the strongest intermolecular force exhibited in the pure substane
A:
Q: ne osmotic pressure as
A:
Q: CH3 HBr H;C CHy CH,COOH ОН
A: Here we have to write the major product and minor products formed in the given reaction . This reac...
Q: Identify the functional group found each of the following molecules.
A: A functional group is an atom or group that determines the chemical behaviour of compound.
Q: Use the Bohr model to calculate allowed energy levels of one-electron atoms Is there an energy level...
A:
Q: (a) How much work is required to compress 4.99 mol of air at 20.4°C and 1.00 atm to one-tenth of the...
A:
Q: Solve the standard entalphies of the following and determine wether the reaction is endothermic or e...
A: 2NH3(g) + 3N2O(g) -----> 4N2(g) + 3H2O(l) CaCO3(s) -------> CaO(s) + CO2(g) C(graphite) + O2(...
Q: QUESTION 8 An aqueous solution of CUNO3 is mixed with an aqueous solution of Nal and a precipitate f...
A: Firstly, let us write chemical reaction and then we can determine solid formed and its colour.
Q: What is the molar mass of a sugar, if a solution of 1.4 g of the sugar in 0.20 L of solution has an ...
A: Please find your solution below : Osmotic pressure is a pressure that should be applied on the solut...
Q: How many moles of lithium hydroxide would be required to produce 57.0 g of Li₂CO₃ in the following c...
A: Answer - According to the question - Given - Chemical reaction - 2 LiOH(s) + CO₂(g) → Li₂CO₃(s) + H...
Q: None of the above
A:
Q: 1. Bronsted-Lowry acid-base reaction between HCIO4 and Br- a. Identify the acid and base in each rea...
A: Two questions based on acid-base reaction that is to be accomplished.
Q: Develop a general equation for the standard heat of reaction as a function of temperature for one of...
A: ANSWER : (a) C4H10(g) →CH2=CH-CH=CH2(g) + 2H2(g)Where:∆H for C4H10 =-125790 J∆H for C4H6 = 109240 ...
Q: ne half-life of
A:
Q: 1. In both the starting material and product the chemical shift of the hydroxyl proton is approximat...
A: Electron withdrawing group and intramolecular hydrogen bond interaction make a proton deshielded and...
Q: An enzyme kinetics experiment is carried out by adding 1.00mg of a 50.0kDa enzyme into a total volum...
A:
Q: Moles of borate present (mol) [Borate] (M) Ksp In(Ksp) 1/T(K-1)
A:
Q: Draw the Lewis structure of PBr,. Include all the lone pairs.
A: Draw Lewis structure of PBr3 ?
Q: How to calculate the mass of 2.7 x 1024 molecules of sugar C12H22O11?
A: Molar mass of C12H22O11 = ( 12 ×12) + (22×1) + (11×16) = 3...
Q: Devise a synthesis of the following compounds starting from benzene.
A: The compound to be synthesized is 5-Bromo-2-nitroaniline. Step 1 Treating benzene with nitrating mix...
Q: What is the molarity of a solution containing 15g sulphuric acid in 150 ml solution? What is the nor...
A: Mass of sulphuric acid = 15 g Volume of solution = 150 ml Molarity of the solution = ? Normality ...
Q: Copper Amax cobalt|Amax copp oncentration, mM 20.00 40.00 60.00 0.177 0.382 0.518 0.2 0.4 0.7 80.00 ...
A: Given : We have to calculate the concentration of cobalt and copper.
Q: Describe Graham's law. Hydrogen chloride gas (HCI) diffuses 1.8 times faster than an unknown gas. De...
A: Graham's law of effusion: Graham's law of effusion states that the rate of effusion of a gas is inve...
Q: An aqueous solution of Ni(N03)2 is mixed with an aqueous solution of KOH and a precipitate forms. A....
A: Nickel Nitrate - Ni(OH)2 is formed as a precipitate in this reaction. The complete reaction is as f...
Q: deviation from Raoult's law, then: a. The mixture will not reach a definite boiling point b. The sol...
A: The correct options are:
Q: This table displays the chemical make-up of human bones. Below, you can see an example of an amino a...
A: Selenocysteine is proteinogenic amino acid. Selenoproteins contain selenocysteine residues. Selenocy...
Q: Calculate the mass and volume (mL) of 5.0 mmol triethylsilane Mw: 116.28 g/mol Density: 0.728 g/mL
A: Given :- moles of triethylsilane = 5.0 mmol Molecular weight = 116.28 g/mol Density= 0.728 g/mL ...
Q: Module 4 Homework Intermolecular Forces: 1. What are the intermolecular interactions between ammonia...
A: Intermolecular forces are the force of attraction that held between two atom. Types of intermolecula...
Q: 50.0mL of 0.100M HCl solution and 50.00mL of 0.100M AgNO3 solution, both initially at 22.54 degrees ...
A: Given, 50.0mL of 0.100M HCl solution and 50.00mL of 0.100M AgNO3 solution, both initially at 22.54 d...
Q: mass percent
A:
Q: II. Analysis. Given below is a schematic diagram for a simple analysis of a novel tetrasaccharide is...
A: D- fructose B - glucose C- galactose A-ribose From TLC profile we can see that four carbohydrate...
Q: Ammonia, NH3, is produced at high temperatures and pressures in the presence of a catalyst as shown ...
A:
Q: 45 Carbonic acid, H,CO, is a diprotic acid and thus, has two K values: K = 4.3 ×10-7; 3 (aq)> a K = ...
A:
Q: Explain why glycated haemoglobin (HbA1C) is a suitable index for long term hyperglycaemia. Briefly ...
A:
Q: Compound Mol. Wt (g/mol) Density (g/mL) b.p. (°C) m.p. (°C) 2-Methyl-2-propanol 74.12 0.786 ...
A:
Q: tert-butyl chloride (2-chloro-2-methylpropane) from tert- butyl alcohol (tert-butanol) using an acid...
A:
Q: Can you suggest a company name for nanomaterials? and a little description of it?
A: Please find your solution below : Nanomaterials are the materials that have at least one dimensions ...
Q: Consider a two-level system of energy separation, 298 cm. At what temperature the population of the ...
A:
Q: A. Standardization of NaOH Solution TRIAL (HCI] Volume Moles Moles Volume [NAOH] HCI HCI NaOH NaOH 0...
A: concentration=moles/volume
Q: The equilibrium constant, K, for the following reaction is 4.68x10-2 at 536 K. PCI5(g) PCI3(g) + Cl2...
A: We have been given equilibrium constant value along with equilibrium concentration at a particular v...
Q: Sodium ethoxide is formed when ethanol (pKa 15.8) is mixed with sodium metal, .Which among the compo...
A: When sodium metal reacts with ethanol, formation of sodium ethoxide and hydrogen gas takes place.
Q: A) FeCl3 CH-CH3 + HCI ČH3 + B) FeCl3 + HCI CH3-C=C-CH2-CH2 –CH3 + H2 Pt, Pd, or Ni C)
A:
Q: Compute the freezing point depression of 0.1% solutions of the following drugs: (a) ascorbic acid; (...
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for yo...
Q: 41. Consider the following reaction: 2 NO2 (g) + C2 (g)→2 NO;CI (g) If 21.0 g of NO2 and 20.0 g of C...
A:
Q: does a co spot on TLC plate have two Rf values?
A: co-spot in TLC
Q: Explain, on the molecular level, how Tin (II) chloride, SnCl2, dissolves in water
A: Hydrolysis A type of chemical reaction which involves the interactions between water and chemicals. ...
Q: For the following molecules, suggest two retrosynthesis pathways with their synthetic equivalents. (...
A: Here we have prepare the following compounds by two retrosynthesis pathway
Q: On a 10 day wilderness expedition you'll need to heat 5.0 kg of water to the boiling point each day....
A: Given, The standard heat of formation of butane at 25°C is - 125.7 kJ/mol 5.0 kg of water to the b...
Complete the balanced dissociation equation for the compound below in aqueous solution. If the compound does not dissociate, write NR after the reaction arrow.
NiS(s)-->
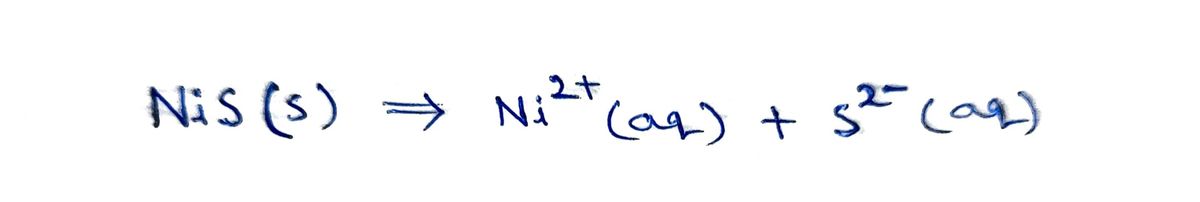
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Complete the balanced dissociation equation for the compound below in aqueous solution. If the compound does not dissociate, write NR after the reaction arrow. HCIO,(aq) – 4- 2- n2+ 3+ D 4+ + 1 3 4 6. 7 8 Os O8 (s) (1) (g) (aq) NR CI Reset • x H,O Delete x H̟O 2 11 3. 2. +Explain how you would separate benzoic acid from NaCl in aqueous solution. Include relevant qualitative test(s) in your explanation.At 77°C, 2.00 mol of notrosyl bromide, NOBr, placed in a 1.00-L flask dissociates to the extent of (9.50x10^0)%; that is, for each mole of NOBr before reaction, (1.000-((9.50x10^0)/100)) mol NOBr remains after dissociation. Calculate the value f Kc for the dissociation reaction 2NOBr(g) ↔ 2NO(g) + Br2(g) Express your answer to three significant figures.
- Calculate the ratio of dissolved benzoic acid to benzoate ion that will exist in solution at equilibrium at a pH of 2.00. The acid equilibrium constant (K a ) for benzoic acid is 6.46 * 10 ^ (- 5) . Note that this calculation doesn't account for the benzoic acid that has precipitated from solution, but you can assume that the higher the ratio, the more benzoic acid will precipitate. Then, Carry out the same calculation for a pH of 4.00, and explain why it was important to reduce the pH to below 4 in this experiment.Complete the balanced dissociation equation for the compound below in aqueous solution. If the compound does not dissociate, write NR after the reaction arrow. 9 X Mg(OH)2(s) → > Co the balanced dissociation eq for the com und below in aqueous solution. If the compound does not dissociate, write NR after the reaction arrow. 10 L C6H3O2(1) – >The value of K, for ethylamine, C,H;NH, is 4.30×104. Write the equation for the reaction that goes with this equilibrium constant. + It is not necessary to include states such as (ag) or (1). +
- Calculate the [H+] and the pH of a buffer solution that is 0.20 M in HC2H3O2 and contains sufficient sodium acetate to make the [C2H3O-2 ] equal to 0.10 M. ( Ka for HC2H3O2 = 1.8 * 10-5)Yeni... O Ara Posta Gonderileri Gözden Geçir Görünüm Yardim 4- Write equations for the acid base reactions that would occur (if any) if ethanol were added to solutions of each of the following compounds In each reaction, label the stronger acid, the stronger base, and so forth.(Please consult the table given below) Ya) NaNH (c) (d) NaOH EGO 16 A 12 IA 17 & 5 8 K(a) Given that Ka for acetic acid is 1.8 x 10-5 and that forhypochlorous acid is 3.0 x 10-8, which is the stronger acid?(b) Which is the stronger base, the acetate ion or the hypochloriteion? (c) Calculate Kb values for CH3COO- and ClO-.
- Using the table of the weak base below, you have chosen Ethylamine as your weak base in the buffer solution. You have already added enough of the conjugate acid salt to make the buffer solution concentration at 0.52 M in this salt. The desired pH of the buffer should be equal to 10.1. Values of K, for Some Common Weak Bases Conjugate Acid Name Formula NH3 CH;NH2 C2H§NH2 CgH;NH2 C3H;N NH,+ CH;NH3* CH;NH3* CH;NH;* C3H;NH+ 1.8 × 10-5 4.38 × 10–4 5.6 × 10-4 3.8 × 10-10 Ammonia Methylamine Ethylamine Aniline Pyridine 1.7 x 10-9 3. Compute the concentration of the weak base in Molarity. (Write your answer in 4 decimal places without the unit).Using the table of the weak base below, you have chosen Pyridine as your weak base in the buffer solution. You have already added enough of the conjugate acid salt to make the buffer solution concentration at 0.62 M in this salt. The desired pH of the buffer should be equal to 4.5. Values of K, for Some Common Weak Bases Conjugate Acid Name Formula Ammonia Methylamine Ethylamine Aniline NH3 CH;NH2 CH;NH2 CH;NH, C;H;N NH,+ CH;NH;* CH$NH;* CH;NH;* C;H;NH* 1.8 × 10-5 4.38 x 10-4 5.6 x 10-4 3.8 x 10-10 1.7 x 10-9 Pyridine 3. Compute the concentration of the weak base in Molarity. (Write your answer in 3 decimal places without the unit).9) a) Label each species as an Bronsted-Lowry acid or base. Show the conjugate acid-base pairs. HCO; + H;O* H¿CO; + H2O (aqueous solution) b) Phenol (HC&H50) is a weak acid (Ka= 1.5 x 10*). Write the equilibrium chemical equation, equilibrium constant expression, and calculate the pH of a 0.25 M solution of phenol. 10) Blood is mainly buffered by HCO; /H2CO; (H2CO3, Carbonic Acid, K= 4.4x10 -") buffer system. Calculate the pH of human blood for a carbonic acid concentration of 0.091M and HCO; concentration of 1.00M.



