Q: Choose (please circle) the most effective buffer at pH 4.75 from the following. 1. 0.20M sodium…
A: Buffer solutions play a crucial role in maintaining stable pH levels in various chemical and…
Q: The following table contains melting temperature (K) and self-diffusion activation energy data for…
A: The given table contains melting temperature (K)and self-diffusion activation energy data for…
Q: plete the following dislocation reactions: a/2[111] + a/2[1-1-1] --> a/6[11-1] + a/3[112] -->…
A: Given the dislocation reactions:a/2[111] + a/2[1-1-1] -->>a/6[11-1] + a/3[112]…
Q: Give the aldol reaction starting material that has a six-membered ring and is needed for the…
A: The aldol reaction is a carbon–carbon bond-forming reaction that occurs in organic chemistry. It…
Q: For each chemical reaction in the table below, decide whether the highlighted reactant is a…
A: Information about the question
Q: Ammonia has been studied as an alternative "clean" fuel for internal combustion engines, since its…
A: Given,Ammonia ( NH3 ) in the combustion with O2 forms nitrogen gas and water vapor.Initial,moles of…
Q: Calculating the pH of a weak base titrated with a strong acid 0/5 Izabella An analytical chemist is…
A: pH = 1.75Explanation:Given:…
Q: b. NO2 + 3H2 Pd 3 atm 2
A:
Q: Is the following alkene s-cis ors-trans?
A: Geometrical isomerism is also called cis-trans isomerism, in which the different arrangements of…
Q: NaOH CH3CH2OH H H3C H NaOH CH3CH₂OH
A: The question aims to find out the major organic products of the given organic reaction with a…
Q: Bromine monochloride is synthesized using the reaction Br2(g) + Cl2(g) 2 BrCl(g) Kp = 1.1 x 10 at…
A: Given ,Reaction : Mass of Br2 = 1.100 Kg = 1100 g Mass if Cl2 = 1.053 Kg = 1053 g We have to…
Q: A major HOH 人 ? minor B C D Η major 人 人 ΟΗ Хон major minor minor
A: SN1 and E1 Reaction : The SN1 and E1 reactions are unimolecular processes. This is a two-step…
Q: What size combinatorial library could be constructed using the substituents and diversity sites in…
A: The objective of this question is to identify substituents and diversity sites and calculate the…
Q: What is the product of the following reaction? HF C14H20
A: Given reaction refers to the replacement of an aromatic proton with an alkyl group. This is done…
Q: The moment of inertia of a CH4 molecule is 5.27 x 10-47 kg m². What is the minimum energy needed to…
A: The moment of inertia of a CH4 molecule = 5.27 x 10-47 kg m2We have to find the minimum energy…
Q: Calculate the pH of a solution made by dissolving 1.410 g of NaOH in 150.0 mL. of water. pH =
A: PH= 13.37Explanation:If you have any problem let me know here in comment box for this thankyou.
Q: Draw the structure of the major organic product of the reaction. Ex CI 1. LiAlH4, ether 2. H3O+
A: Lithium Aluminium Hydride (LAH) reduces most of the functional groups present in organic compounds…
Q: When 2,2-dimethylcyclohexanol is treated with acid, 1,2-dimethylcyclohexene and iso-…
A: The given reaction is an acid-catalyzed dehydration of 2, 2-dimethylcyclohexanol.Explanation:Step…
Q: Question 12 Draw the appropriate Newman projection which leads to the major E2 ELIMINATION product.…
A:
Q: Hu Hd 12 10 8 6 4 2 0 0 4 8 12 16 20 24 28 32 36 Volume Base Added (mL) 8A. What was in the…
A: A titrant is the solution which is added from burette while a sample is taken in titration flask.In…
Q: What carbohydrate is shown below based on the Fischer projection?
A: Epimers are stereoisomers in which two isomers have more than two chiral centers and the…
Q: Fill in the left side of this equilibrium constant equation for the reaction of hypochlorous acid…
A: Dissociation of HClO in water is given by:
Q: 9) Consider the following reaction: what is the expected product? NH₂ Br₂, H₂O ?
A:
Q: Draw the major organic product(s) of the following reaction. CI + NaOCH3 CH₂OH
A:
Q: In the laboratory you dissolve 17.3 g of zinc fluoride in a volumetric flask and add water to a…
A: The objective of the question is to calculate the molarity of the solution, the concentration of the…
Q: Do all titrations of a strong base with a strong add have the same PH at the equivalence point?
A: The question is asking whether the pH at the equivalence point of a titration between a strong base…
Q: The following molecule is formed in an intramolecular aldol condensation reaction. Draw the organic…
A: The organic starting material of the reaction is 3,5-dimethylheptanedial.Explanation:
Q: Need help
A: The objective of the question is to give an educated guess about the major product of the nitration…
Q: Which statement describes why aldol reactions with ketones are low yielding? A.Ketones do not have…
A: In an aldol reaction, a carbonyl compound (aldehyde or a methyl ketone) reacts in the presence of a…
Q: The preparations of two aqueous solutions are described in the table below. For each solution, write…
A: Given,0.6 mol of HCl is added to 1.0 L of a 0.4 M NH3 solutionacids:bases:other:0.08 mol of HBr is…
Q: Draw the structure of the self-aldol addition product that would form from propanal. Don't forget to…
A: Have a look to the explanation..Explanation:Step 1:Deprotonation of aldehyde. Step 2: Condensation…
Q: Substitution and elimination: predict the product Maximum allowed tries per question: Unlimited (7)…
A: The objective of the question is to predict the product formed in the following reaction given.
Q: 5- Explain why the -NHCOCH3 in acetanilide is only moderately activating while the -NH2 group in…
A: Given question is based on electrophilic aromatic substitution reaction.Note: According to Bartleby…
Q: Is there a pattern to change in pressure, amount, K, pH?
A: While general patterns exist, the precise impact of variations in pressure, amounts, equilibrium…
Q: H3C- CH3 Na2Cr2O7 H2SO4
A: Toluene is oxidized by acidified permanganate or dichromate into benzoic acid. This is because a…
Q: Propose a concise and efficient synthesis of the target molecule. Br steps starting material C…
A:
Q: Draw both additional resonance structures for the allyl -harr longleftrightarrowradical below.…
A: In the given radical species there are two consecutive double bonds. So the radical takes part in…
Q: NOTE: Your only sources of carbon in the final product are the starting materials on the left. It is…
A:
Q: Using the priority rule, identify the configuration at each chiral center: Please type one of the…
A:
Q: When the following half reaction is balanced under acidic conditions, what are the coefficients of…
A: The objective of the question is to balance the given half reaction under acidic conditions and…
Q: Identify the Major and ALL Minor product(s) that are expected for each of the following reactions.…
A: Steps to proceed with the reaction: As Br is attached with a secondary carbon atom, the reaction may…
Q: Select the correct IUPAC name for the cycloalkane:
A: The IUPAC nomenclature provides a set of rules by which an organic compound can be named…
Q: Draw the structure of the major organic product for the following reaction:
A: Carbonyl compounds react with a secondary amine to form an enamine. Enamines are nitrogen analogous…
Q: 1 7. What are the stereochemical designations for the two chiral centers 1 and 2 of the following m…
A: R and S configurations are different ways to spatially arrange atoms around a chiral center. R comes…
Q: 60 40 1 4 20- 3 0 000 2 9 10 19 6 11 14 7 8 13 5 12 3000 2000 1000 650 Wavenumber [cm-11
A: The question aims to identify the unknown alcohol by using the given IR spectrum and its boiling…
Q: Draw major E2 ELIMINATION product. Hint: Review Chapter 10.3. the elimination can only occur if the…
A:
Q: Draw the following compounds by representing them as covalent bonds using VSEPR Theory: a. CO2 b.…
A: "Since you have asked a question with multiple sub-parts, we will answer the first three sub-parts…
Q: Mass of CaCO3 standard (g) Volume of CaCO3 used (mL) Volume of EDTA Trial 0.15099 34.90 Trial 2…
A: The % mass of a molecule in a given mixture can be calculated using the following equation-
Q: 1. (a) Compound A,B and C are isomers with molecular formula of C4H8O. When compound A,B and C…
A: The objective of the question is to deduce the structures of compounds A, B, and C based on their…
Q: calculate the pH of a 0.70 M solution of KOH
A: Given that,The concentration of strong base,pH of the solution = ?
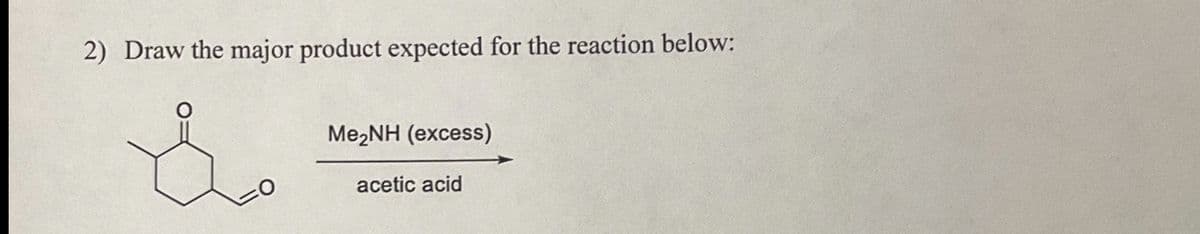
Step by step
Solved in 3 steps with 5 images

- Draw the products of the three step reaction sequence shown below. Ignore inorganic byproducts. If the reaction results in a mixture of ortho and para isomers, draw only the para-product. Select to Draw NO₂ 1. LiAlH4 2. H₂O* Cl₂ AICI 3 Select to Draw CH3C(O)CI Select to Drawpossible products of the reaction belowa)draw the products of the reaction below
- Which product would likely be the major product for the following reaction?a. Bb. Cc. Ad. DWhich product would likely be the major product for the following reaction?a. Ab. Cc. Dd. B3. Prepare each compound from cyclopentanol. More than one step may be needed.llustrate the mechanism of the reaction. Include the intermediate product and by-product formed in the reaction. (see attached photo)
- 5..8. What is the expected major for the following reaction ?4. propose a mechanism for the following reaction (don't go over 18 electrons!). be sure to use curved arrows, state the electron count of each intermediate, and name the reaction at each step.Which product would likely be the major product for the following reaction?a. Cb. Dc. Ad. B
- Draw all the potential products from the reaction below.Choose the best reagents to complete the following reaction. There's another answer choice not shown on screen: E) 1. CH3Li 2. H3O+ Hint: The correct answer is not option C.4.Draw all of the target products for the reactions below. Identify the major product



