
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 5.9B, Problem 5.15P
Draw three-dimensional representations of the following compounds. Which have asymmetric carbon atoms'? Which have no asymmetric carbons but are chiral anyway? Use your models for parts (a) through (d) and any others that seem unclear.
- a. CIHC=C=CHCI 1,3dichioroprop8diene
- b. CIHC=C=CHCH3 1-chlorobuta-1,2-diene
- c. CIHC=C=C(CH3) 1-chloro-3-methy1buta-1,2-diene
- d. CIHC=CH—CH=CH2 1-chlorobuta-1,3-diene
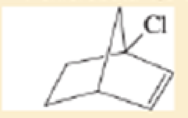
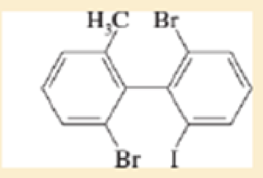
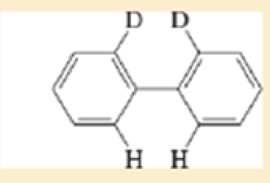
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Consider the Newman projections, A-C.
CHO
Н.
H
CHO
OH
A
CH₂OH
OH
но.
н
H
ОН
CH₂OH
B
н.
НО
CHO
OH
C
.CH₂OH
н
i. Determine the structural relationship of A and B; B and C.
ii. Without a change in conformation, assign the configuration to the chiral centers in B.
5. What are the structures of A, B, and C?
NBS
NaOCH2CH 3
A
B
hv
CH3CH,OH, 4
CHO
CH2=CHCO,H
1. 03
2. S(CH3)2
HO2C
CHO
Draw a structural formula of the S configuration of the compound shown below.
• Use the wedge /hash bond tools to indicate stereochemistry where it exists.
Include H atoms at chiral centers only.
• Ifa group is achiral, do not use wedged or hashed bonds on it.
CH3
CH3
CH;CHCHCN
CH,CH,CH,CHCH,CH,
CH2NH2
Draw a structural formula of the RS configuration of the compound shown below.
Use the wedge /hash bond tools to indicate stereochemistry where it exists.
• Include H atoms at chiral centers only.
If a group is achiral, do not use wedged or hashed bonds on it.
ÇI
Chapter 5 Solutions
Organic Chemistry (9th Edition)
Ch. 5.2 - Determine whether the following objects are chiral...Ch. 5.2A - Prob. 5.2PCh. 5.2B - Prob. 5.3PCh. 5.2B - Prob. 5.4PCh. 5.2C - Prob. 5.5PCh. 5.3 - Prob. 5.6PCh. 5.3 - Prob. 5.7PCh. 5.4D - Prob. 5.8PCh. 5.4D - Prob. 5.9PCh. 5.4D - Prob. 5.10P
Ch. 5.5 - Prob. 5.11PCh. 5.7 - When optically pure (R)-2-bromobutane is heated...Ch. 5.7 - Prob. 5.13PCh. 5.8 - Prob. 5.14PCh. 5.9B - Draw three-dimensional representations of the...Ch. 5.10A - For each sot of examples, make a model of the...Ch. 5.10A - Draw a Fischer projection for each compound....Ch. 5.10B - Prob. 5.18PCh. 5.10C - For each Fischer projection, label each asymmetric...Ch. 5.11C - Prob. 5.20PCh. 5.13 - Prob. 5.21PCh. 5.13 - Prob. 5.22PCh. 5.15 - Prob. 5.23PCh. 5.16A - Prob. 5.24PCh. 5 - The following four structures are naturally...Ch. 5 - For each structure, 1. star () any asymmetric...Ch. 5 - Prob. 5.27SPCh. 5 - Prob. 5.28SPCh. 5 - Prob. 5.29SPCh. 5 - Prob. 5.30SPCh. 5 - Prob. 5.31SPCh. 5 - Prob. 5.32SPCh. 5 - Prob. 5.33SPCh. 5 - Prob. 5.34SPCh. 5 - For each structure, 1. draw all the stereoisomers....Ch. 5 - Prob. 5.36SPCh. 5 - Prob. 5.37SPCh. 5 - 3,4-Dimethylpent-1-ene has the formula...Ch. 5 - A graduate student was studying enzymatic...Ch. 5 - Prob. 5.40SPCh. 5 - Prob. 5.41SP
Additional Science Textbook Solutions
Find more solutions based on key concepts
Determine [OH], [H+], and the pH of each of the following solutions. a. 1.0 M KCl b. 1.0 M KC2H3O2
Chemistry
Calculate the lattice energy of CaCl2 using a Born-Haber cycle and data from Appendices F and L and Table 7.5. ...
Chemistry & Chemical Reactivity
Fully developed conditions are known to exist for water flowing through a 25-nim-diameer tube at 0.01 kg/s and ...
Fundamentals of Heat and Mass Transfer
2. Why shouldn’t you work in a laboratory by yourself?
The Organic Chem Lab Survival Manual: A Student's Guide to Techniques
4. 38 Strontium has four naturally occurring isotopes, with mass numbers 84, 86, 87, arid 88.
a. Write the atom...
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
Q1. What is the empirical formula of a compound with the molecular formula
Chemistry: A Molecular Approach
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- online.butiercc.edu/Courses/ 147954 2493001/ take zınb Question 4 1 pr Which molecule is the enantiomer of this molecule? H. H3C Br CICH, CH3 Bri C H CH,CI CH,CI Br C-H CH3 H3C -Br CICH, H H3C CICH, Brarrow_forwardTwo structures are shown. Identify the asymmetric carbons in each structure, classify as chiral or achiral, and identify the number of stereocenter(s). H. „CH3 H3C CH3 H H. CH3 compound A compound B Which image shows the asymmetric carbon(s) in compound A highlighted in red? CI H H3C CH3 H,C CH3 O Neither. There are no asymmetric carbons in compound A. H. H3C -CH3 C H3C CH3 Compound A is . There are stereocenters in compound A. Which image shows the asymmetric carbon(s) in compound B highlighted in reď? .CH3 H H. CH3 H3C.C H O Neither. There are no asymmetric carbons in compound B. Compound B is There are stercocenters in compound B. *Please explain all the questionsarrow_forwardDraw a structural formula of the R configuration of the compound shown below. ÇNH₂ CN • Use the wedge/hash bond tools to indicate stereochemistry where it exists. • Include H atoms at chiral centers only. • If a group is achiral, do not use wedged or hashed bonds on it. #[ ] در ?arrow_forward
- 3: Draw bond-line structures (with wedges or dashes, where appropriate) of each of the following molecules. Be sure to draw the correct stereoisomer of any chiral molecules. a: H. H I I C: H. OH b: CH3 H S CH3 H Br CH3 CH3 CI CH3arrow_forwardDraw a structural formula of the RS configuration of the compound shown below. OH R U CH 8 _CH_ SI ephedrine [*** NH-CH3 CH3 • Use the wedge/hash bond tools to indicate stereochemistry where it exists. • Include H atoms at chiral centers only. • If a group is achiral, do not use wedged or hashed bonds on it. ▾ Sn [F ?arrow_forwardTwo structures are shown. Identify the asymmetric carbons in each structure, classify as chiral or achiral, and identify the number of stereocenter(s). H. „CH3 H3C CH3 H H. CH3 compound A compound B Which image shows the asymmetric carbon(s) in compound A highlighted in red? CI H H3C CH3 H3C CH3 O Neither. There are no asymmetric carbons in compound A. H3C -CH3 H3C CH3 Compound A is . There are stereocenters in compound A. Which image shows the asymmetric carbon(s) in compound B highlighted in red? CH3 H H. CH3 H3C.C H O Neither. There are no asymmetric carbons in compound B. Compound B is . There are stercocenters in compound B. *Please explain all the questionsarrow_forward
- 4. For each of the following molecules, label each stereocenter as R or S. Label the molecules appropriately as chiral or meso. a. b. methionine NH₂ H d. но. OH c. D = 2H, T = ³H, isotopes of hydrogen. f.T. OH OH CI Y ОНarrow_forward2. Indicate all stereogenic carbons, assign absolute configuration (R or S) and determine if the molecule is chiral or achiral. Br H. HOll.. CI CH3 Br CI H. d) Br H. H. f) H3C. NH2arrow_forward1. Redraw the structure below, showing both nonaromatic rings in chair conformations. Place the bridgehead hydrogen atoms in axial positions. Pay special attention to drawing the correct stereochemistry for all chirality centers, as shown in this structure. Assign each stereocenter as R or S. „OCH3 "OH OH H2Narrow_forward
- Does the molecule below exist as a pair of enantiomers? If so, change the bonds to wedges and dashes to reflect S stereochemistry. If the molecule does not exist as a pair of enantiomers, check the box below. H₂C- HO Does not exist as enantiomers X :0 Stmarrow_forwardwhat is the structure and jvalues? (c10H14O).arrow_forward2. Explain with a couple of acyclic or cyclic examples (5 carbons or more) what is crit- ical for you when establishing whether a molecule is chiral or acral.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License