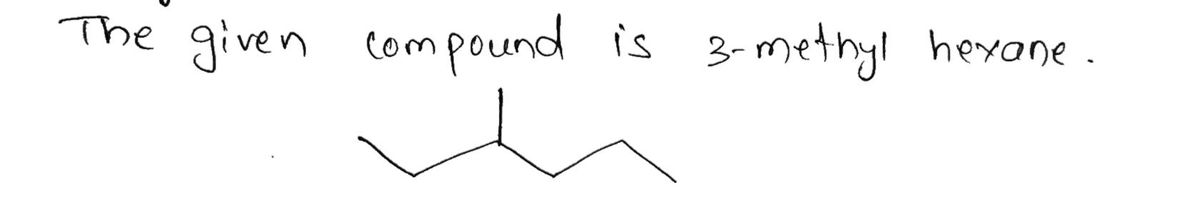
Which of the following is the least stable Newman Projection for 3-methyl hexane looking down the indicated bond (C3-C4 bond)? H H. H CH₂CH3 H H HỊCH, CHỊCH H r H CH₂CH3 H CH3 CH2CH3 -H CH₂ CH3 CH₂CH₂
Q: The reaction AB is first order in [A]. Consider the following data. Time (s) [A] (M) 0.0 3.0 10.0…
A: The integrated rate law for a first order reaction is given by- ln[A]=−kt + ln[A]o , where where…
Q: 44. The reaction of 1-bromo-2-fluorobenzene with furan in the presence of one equivalent of Mg gives
A: given :
Q: ||| Identifying phase transitions on a heating curve A pure solid sample of Substance X is put into…
A: Melting point is the temperature at which a solid substance changes state from a solid to a liquid.…
Q: 3) How many liters at STP would 3.45 moles of CO occupy?
A: Given that number of moles of CO = 3.45 mol STP conditions are Temperature = 0 °C = 273.15 K P = 1…
Q: result for letter D please.
A: Given electronic configuration is d) 1s22s22p63s23p64s23d
Q: Can you help me number 6? Is this correct answer with show my work including the Significant Figures…
A:
Q: What is the correct way to write mercury in a formula? How would it be used in the examples and why…
A: Mercury ions can exist in two oxidation states : +1 and +2. Mercury (II) ions are written as Hg2+.…
Q: What is the molar solubility of AgCl (Ksp = 1.80 x 10:10) in 0.480 M NH? (Kf of Ag(NH₂)₂* is 1.7 x…
A:
Q: Consider the following reaction: PCI (9) PCs (g) + Cl₂(g) An equilibrium mixture of the three gases…
A:
Q: What's 12,13 and 16?
A: To calculate the mole fraction, mole percent and ppm of a saturated solution of NaOH at 20°C.
Q: Decide how the sketches below would be listed, if they were listed in order of decreasing force…
A:
Q: what is the product and mechanism for this reaction?
A: Good leaving groups are weak bases. They’re happy and stable on their own. Some examples of weak…
Q: Draw the products of the following reactions: only ng di (a) H,CCN d+ + bha bon 161 not to be an H,C…
A:
Q: If 0.70 g of FeCl3 is used to iodinate 8.42 g of benzene with sufficient ICl, what is the average…
A: According to the question, The mass of FeCl3= 0.70 gThe mass of benzene = 8.42 gThe molar mass of…
Q: 4) a) If 3.40 moles of NaNO, was dissolved into 2.58 L, what is the molarity? b) If you measured…
A: Consider the given information as follows; (a) Moles of NaNO3 = 3.40 mol Volume = 2.58 L…
Q: Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product…
A: Arrow pushing or electron pushing is a technique used to describe the progression of organic…
Q: 3. Draw a molecule with the molecular formula C6H10O that would have the following peak(s) in its IR…
A: We have to determine the structure of the compound on the basis of given IR data
Q: Methane and chlorine react to form chloroform and hydrogen chloride, like this: CH4(9)+ 3Cl₂(g) →…
A: According to Le chateliar's principle, the reaction at the equilibrium shifts in a direction that…
Q: Lewis dot structures have three general exceptions to the octet rule, one of which is? Molecules in…
A:
Q: 5) 15.0 grams of Mg(NO3)2 contains how many representative particles? (Molar mass of Mg(NO3)2 =…
A:
Q: Which polymer best corresponds to the IR spectrum? 100 ความ 4000 3000 O Teflon www PVC (polyvinyl…
A: The principle of IR (infrared) spectroscopy is based on the fact that different chemical bonds in a…
Q: (9) Given the reaction mechanism below, write the net (balanced) reaction and identify any…
A:
Q: 7. Complete the following table. element period Be He electron configuration 15²25² [Ne]3s Look at…
A: Prediction of Period from electronic configuration : In the electronic configuration , the…
Q: a. b. Br HOH NaOEt EtOH H₂SO4 heat
A: The above given reactions are example of elimination reaction in which the final product is an…
Q: Ether and hexane are considered to non-polar solvents, in all honesty, ether is only slightly,…
A: Introduction The Rf value (retention factor) in thin-layer chromatography (TLC) is a measure of the…
Q: Consider the following reaction: CO₂ (9)+CCL (g) 2COC (9). Calculate A, G for this reaction at 125 C…
A:
Q: 2. Brz Y FeBr3 A NH3 NaNH, B CH3CI AICI 3 D с HNO3 (x's) H₂SO4 (x's) H₂50, (x's)
A: Aromatic compounds undergo electrophilic substitution reactions like halogenation, alkylation,…
Q: Draw a structural formula for the major organic product of the reaction shown below. Br CH3C=CH₂ +…
A:
Q: Using the mass spectrum below select the most likely fragment peak at 57: Relative Intensity 100 80…
A: We need to find the molar mass of each of the options given. mz=molar mass of the fragmentcharge of…
Q: O Ligh Home Tren APTENT PRAT
A:
Q: 2. Predict the product in the following reaction and show a mechanism for its formation: Но H₂C- A…
A: This is an example of E1 elimination reaction .The initial step is formation of a carbocation…
Q: 3. In addition to vivid colors, the fireworks also use other visual features such as smoke, glitter…
A: In the given question, we have to write the ionic formula of the following compound, writing the…
Q: 0 2 33 give products of this reaction and outline the meachanisms M₂0 Br CI Na OH 350% NaOH 350°C Na…
A: This is simple question of nucleophilic aromatic substitution reaction where the nucleophile is…
Q: 55.8 % C, 11.6% H and 32.6% N by mass Determine the molecular formula of the particles of the…
A: The mass percentage of C, H, and N is 55.8%, 11.6%, and 32.6%, respectively. The mass percentage…
Q: DIRECTION: Fill in the missing parts of the table below. 1-2 3 4-7 8-10 11-13 14-17 18-20 ACID HNO2…
A: Given incomplete acid base reaction
Q: Give the structure, using the representation shown is parentheses, for each of the following…
A:
Q: Explain your choice, specifically describing the role of each metal in the redox process. A complete…
A:
Q: Fill in the missing symbol in this nuclear chemical equation. 38 19 K 38 18 Ar +
A: Check the type of the given nuclear reaction and write down the missing symbol in the box.
Q: Endothermic Possible answers No Answers Chosen A reaction with unstable products and stable…
A: Endothermic reactions absorb heat from their surroundings, while exothermic reactions release heat…
Q: How many oxygen atoms are on the right-hand side of the following chemical equation? Express your…
A:
Q: Use the VSEPR model to predict the bond angles about each numbered atom. H ++ The predicted angles…
A:
Q: 271 Question out of the described bellow, which following graphs one of is NOT linear for the 2 of…
A: Given that, for the disappearance of the reactant A, we have to tell which one of them is not…
Q: e compound iron (II) chloride, FeCl₂ is soluble in water. Write the formulas for the ions that…
A: when ionic compound dissolves in water it gives positive and negative ions
Q: Show how you can carry out the following transformations with the limitations given below and any…
A: There are 2 steps in following conversion - 1) First step is nitration . Nitration in presence of…
Q: The table below lists information about the radioactive decay of three nuclides. Fill in the missing…
A:
Q: Write the condensed structural formula and the name of the oxidized product when the following…
A: The product of given reactions can be explained as:
Q: What is the density of a metal if a 25.0g piece of a metal causes the water in a graduated cylinder…
A:
Q: Can you help me number 2? Is this correct answer with show my work including the Significant Figures…
A:
Q: 2. For the reaction of A + 2B → AB2, the following date are obtained: INITIAL [A] INITIAL [B] 0.660…
A: Calculation of rate of the reaction: Let the order of the reaction with respect to [A] be x and…
Q: An iron kettle weighing 1.49 kg contains 2.82 kg of water at 29.1°C. The kettle and water are heated…
A: The total amount of heat/energy absorbed or lost by any body is expressed by Q = mc∆T m= mass of the…

Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Which Newman projection demonstrates the least stable conformation of the C3-C4 bond of 3-methylhexane? A) C2H5C2H5 H3C H B) C3H7 C3H7 H TH H 'H C) D) H. Н' H, I I C3H7 .H CH3 C3H7 C₂H5 C₂H5 H CH35 6 3 4 2 Note the root associated with each of the following number of atoms: Example Root Single # of Carbons 1 anes double enes anes Single triple bend nes anes single anes single H CH4 H X H HTC1 1C1 H H-C CCC H H H H H H H HIC CIG C-H HIC H H H H -H CIH HIC -C-C H H H double bond Alkynes H с -H Name Methane C래니 сгни Ethene H H H -C-H H H H H H—C—C C C C C-H H H H H H H2) Label the least favorable conformation and most favorable conformation of the substituted cyclohexane below. Explain your choices in 1-2 sentences or less. H3C H3C H3C F OH CH3 CH3 H3C CH3 F CH3 OH H3C CH3 H3C снаанз F CH3 CH3 -OH
- Rank the following Newman projections in order of decreasing energy, from least stable to most stable. H₂C H H A H ››› > > > x H₂C H H B H H₂C H H H C H H₂C H H D 8 M olo Ar 8. K2. Draw the Newman projection of the following compounds about the indicated bonds: F, CH3 H CH3 H OH H H CH3 Cl. НО HWhich of the species contains a r bond? co3- 03 HCN H,0 Which of the species contains a delocalized a bond? H,0 HCN O Co-
- The following are all accurate Newman projections for compound X looking down the highlighted bond, but in different conformations. Which of the shown conformations is the lowest energy conformation of X? H. H CH3 CH3 (A) C(CH3)3 H (H3C)3C- H (B) Η, Η снанз X H3C (H3C)3C H3C (C) H H H. H3C H H3C (D) C(CH3)3 HWhich Newman projection corresponds to the compound below: H3C CI OA ОВ O C OD 20 A CH3 CH3 H H F4 H3C H F5 B CH3 CH3 H CI H Me H3C CI CI C H CH3 MacBook Pro F7 H CH3 H3C H DII FB H H .H CI Nex F9Account for the fact that among the chlorinated derivatives of methane, chloro- methane has the largest dipole moment and tetrachloromethane has the smallest dipole moment. Molecular Formula Dipole Moment (debyes, D) Name CH,CI CH,CI, Chloromethane 1.87 Dichloromethane 1.60 Trichloromethane CHCI, 1.01 Tetrachloromethane CCI,
- For each example, specify whether the two structures are resonance contributors to the same resonance hybrid. H. :0-CH2 а) HO-CH2 CH2-CH2 CH2 CH2 yes no ö: :ö: b) H2C=CH-C-H H2C-CH=ċ-H c) H2C=CH-CH-CH3 H2C-CH=CH-CH3What is the most stable chair conformation of the 4-methoxy-1,2- dimethylcyclohexane structure given below? A a B b с с D d OCH: "CH3 H3C 4-methoxy-1,2-dimethylcyclohexane OCH: OCH3 H3C H3C- H3C- OCH3 H3C -OCH3 ĆH: CH3 CH3 CH3 A B DThe first resonance structure of acetamide, CH3 CONH2, is shown. H :0: ☐ || H-C-C-N-H H H Determine the correct second resonance structure of acetamide. H :0: -1 +1 H-C-C-N-H H H H :0: -1 ☐ ☐ H-C=C-N-H H H H :0: H-C-N-C-H HH ●● -1 H:0: H H-C-C=N—H H H

