A Diels–Alder reaction calls for the use of 5.8 mL5.8 mL of a 4.0 M solution of cyclopentadiene in methanol. Calculate the number of moles of cyclopentadiene present in this volume.
Q: Give two different ways to prepare the following compound by the Diels-Alder reaction. Explain which…
A: To find: The two different ways to prepare the given compound by diels alder reaction.
Q: Draw a structural formula for the Diels-Alder adduct formed by reaction of each diene and dienophile…
A: In a chemical reaction; the substance which involves in conversion is said to be reactant whereas…
Q: Predict the product for the following Diels-Alder reaction. сосн,
A: We have given the organic reaction and we have to find the major product of the reaction.
Q: 2,3-Di-tert-butyl-1,3-butadiene is extremely unreactive in Diels–Alder reactions. Explain.
A: Diels Alder reaction is the reaction between a diene and a dienophile.
Q: The ratio of reactivity of tertiary:secondary:primary hydrogen atoms in a bromination reaction at…
A:
Q: Calculate the theoretical yield for the diels alder reaction? The data is: Weight of…
A:
Q: What is the purpose of A Pericyclic Diels-Alder Reaction ?
A: Diels-Alder reaction is a common pericyclic reaction between a conjugated diene and a dienophile,…
Q: Calculate the theoretical yield, in gm, of product that can be produced using 10 gm of anthracene…
A: Diels–Alder reaction is a chemical reaction between a conjugated diene and a substituted alkene,…
Q: In your experiment, Na2CO3 and K2CO3 were differentiated by flame test. describe the expected…
A: A flame test is an analytical procedure used in chemistry to detect the presence of certain…
Q: Arrange the following dienes in order of increasing reactivity in a Diels-Alder reaction by typing…
A: In Diels alder reaction, a diene will react with an enophile. The diene in the Diels alder reaction…
Q: calculate and state the molecular weight of a Diels-alder product resulting from the reaction of…
A: Diels-Alder reaction: The Diels–Alder reaction is a pericyclic reaction (cycloaddition) between a…
Q: explain the reaction and mechanism of Diels alder reaction
A:
Q: CH3 +
A:
Q: Why methanol is a good solvent for UV but not for IR?
A:
Q: Arrange the following dienes in order of increasing_reactivity in a Diels-Alder reaction by typing…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Indicate the structures of the dienes and phyllodes that give rise to the Diels-Alder adducts:
A: Diels alder reaction is an example of pericyclic reaction in which diene and dienophile is involved.
Q: [References) Draw structural formulas for the diene and dienophile that combine in a Diels-Alder…
A:
Q: Explain the principal synthetic value of the Diels-Alder reaction.
A:
Q: Why is the diels alder reaction important? What is a compound that would be not possible to…
A: 1. Diel Alders reaction is important to synthesize cyclic compounds (i.e. cyclohexenes) of different…
Q: Explain why free-radical halogenation usually gives mixtures of products
A: A free radical halogenation reaction always substitutes the hydrogen atom with a halogen atom. It is…
Q: Benzyl alcohol (0.500 mmol) and benzoic acid (0.500 mmol) were produced from the eaction of…
A: Here we are required to find the mass of benzaldehyde needed for the reaction.
Q: COOH benzene reflux COOH
A:
Q: Write the structures of the starting diene and dienophile necessary to prepare each molecule and…
A: This product is type of diels-alders reaction. In this reaction, a conjugated diene and an alkene…
Q: 5. During the Diels-Alder reaction, you had to remove excess maleic anhydride. Which of the…
A: 5. Vacuum Filteration technique could be used to remove excess of Maleic anhydride if the product…
Q: What does it mean if a Diels Adler reaction mixture turns black in the reaction?
A: This question is asking the reason of formation of black solution after the reaction. We know that,…
Q: Why does a [2+2] cycloaddition reaction require a thermal condition to be met for initiation?
A: The reason is as follows:
Q: Could the separation in this experiment have been done in a different order? That is, isolation of…
A: The chemical reaction may be written as: CaCO3 + 2 HCl→ CaCl2 + CO2 + H2O To separate SiO2 first,…
Q: Calculate the theoretical performance and yield percentage of methyl m-nitrobenzoate Amount of…
A: Given: Volume of methyl benzoate taken = 2.00 mL Mass of purified methyl m-nitrobenzoate produced…
Q: Write the equation and the corresponding mechanisms for the Diels-Alder reactions of cyclopentadiene…
A:
Q: Diels-Alder reaction mol amount of anthracene used 0.035g 0.2529 6.2145 amount of maleic anhydride…
A:
Q: A Diels-Alder reaction calls for the use of 5.9 mL of a 4.0 M solution of cyclopentadiene in…
A: Given, concentration of solution of cyclopentadiene = 4.0 M =4.0 mol/L…
Q: Explain why butadiene sulfone was used in the Diels-Alder reaction with maleic anhydride? Why is…
A:
Q: Using the table of bond dissociation enthalpies , calculate ▲H° for bromination of propane to give…
A:
Q: Under certain conditions, 1,3-butadiene can function as both a diene and a dienophile. Draw a…
A: The Diels-Alder reaction is a conjugate addition reaction of a conjugated diene to an alkene or an…
Q: calculate and state the molecular weight of a diene with the molecular formula C10H16. calculate the…
A: The amount of a substance present in a system can be expressed by various units such as grams,…
Q: Calculate the theoretical yield and percent yield of the product formed from. Reaction between…
A: Given - Mass of Tetraphenylcyclopentadienone = 1.005 g Mass of Maleic Anhydride = 1.203…
Q: A Diels–Alder reaction calls for the use of 5.7 mL5.7 mL of a 4.0 M solution of cyclopentadiene in…
A: Molarity (M) of a solution is defined as the number of moles of solute dissolved per volume of the…
Q: Draw structural formulas for the diene and dienophile that combine in a Diels-Alder reaction to form…
A:
Q: Which of the following can be used as the required diene in a Diels-Alder reaction? NO2 II IV а. I…
A:
Q: How to calculate the percent yield of the Diels Alder Reaction. Based on the experimental data of…
A: Since we know that percent yield can be calculated by using the given below formula. Percent yield =…
Q: R .R allylic position benzylic position Benzylic cations, anions, and radicals are all more stable…
A:
Q: If you began an experiment with 5.0 mL of cyclopentadiene and 5.16 g of methyl acrylate, and you…
A: Cyclopentadiene + methyl acrylate--> diels alder 5.0 ml 5.16 g…
Q: From the four following choices, select the best dienophile for a Diels-Alder reaction.
A:
A Diels–Alder reaction calls for the use of 5.8 mL5.8 mL of a 4.0 M solution of cyclopentadiene in methanol. Calculate the number of moles of cyclopentadiene present in this volume.
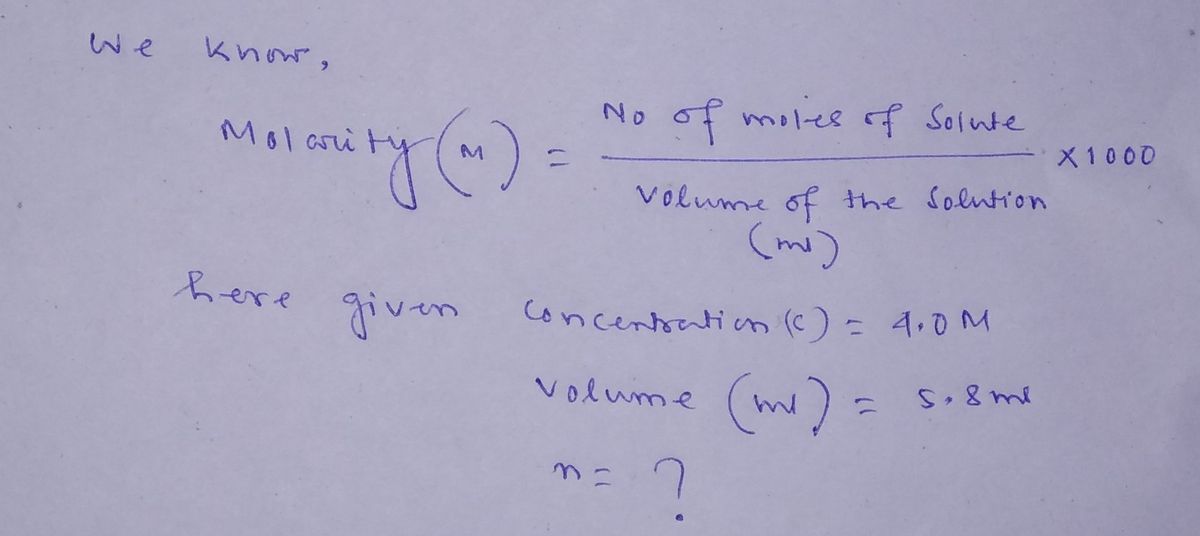
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Born-Haber cycle; I don’t know how to formulate it for this reaction.Write the propagation steps leading to the formation of dichloromethane (CH2Cl2) from chloromethane21.) Calculate the enthalpy of hydrogenation of benzene to cyclohexane from the following reactions A,H (kJ/mol) C6H6 (1) + 15/2 02 (g) → 6 CO2 (g) + 3 H20 (1) C6H12 (1) + 9 02 (g) → 6 CO2 (g) + 6 H20 (1) H2 (g) + ½ 02 (g) → H2O (1) -3268 -3920 -285.83 a.) -205 kJ/mol b.) -1507 kJ/mol c.) -938 kJ/mol d.) -366 kJ/mol
- A Diels-Alder reaction calls for the use of 5.2 mL of a 4.0 M solution of cyclopentadiene in methanol. Calculate the number of moles of cyclopentadiene present in this volume. number of moles of cyclopentadiene: mol * TOOLS x10Comment on the difference between the enthalpy of combustion values of anthracene, napthalene, and phenathrene. delta H anthracene = -7063.8±5.3 kJ/mol delta H napthalene = -5160. ± 20. kJ/mol delta H phenanthrene is -7040. ± 30. kJ/molFree‑radical halogenation can occur with chlorine and a source of direct UV radiation or sunlight. Chlorination of 2,4‑dimethylpentane via radical halogenation leads to the formation of all three of the products shown. Estimate the relative percentages of each product that will be formed using this means of halogenation. Presume that 1 equivalent of chlorine is used.
- Use the following atomic weights and quantities to calculate the overall % yield of tetraphenylcyclopentadienone. Remember that your yield must be based on the limiting reagent, and that grams must be converted to moles. Filling out most of the chart will help. Give only two significant digits in your answer. If after rounding the answer is a whole number, do not include a decimal point. C = 12, H = 1, K = 39, O = 16 benzil + dibenzyl ketone potassium + → tetraphenylcyclopentadienone hydroxide formula formula weight grams 2.314 7.231 3.857 3.173 moles Answer: % yieldWhy is benzene less reactive than hexane in terms of stability of free radicals?Use the following atomic weights and quantities to calculate the overall % yield of tetraphenylcyclopentadienone. Remember that your yield must be based on the limiting reagent, and that grams must be converted to moles. Filling out most of the chart will help. Give only two significant digits in your answer. If after rounding the answer is a whole number, do not include a decimal point. C = 12, H = 1, K = 39, O = 16 formula formula weight grams moles Answer: benzil + dibenzyl ketone + potassium hydroxide tetraphenylcyclopentadienone 4.135 8.646 1.140 % yield 6.956
- write both the thermodynamic & kinetic products for the reaction of: 1,3-pentadiene with HBrThe compound below is treated with chlorine in the presence of light. H₂C CH3 H3C CH₂CH3 Draw the structure for the organic radical species produced by reaction of the compound with a chlorine atom. Assume reaction occurs at the weakest C-H bond. • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms.True or False: Acetylene is a naturally occurring conjugated diene True or False: The Diels-Alder reaction has the stereochemistry of the dienophile is retained in the product. True or False: When looking at kinetic vs. thermodynamic products the kinetic product predominates at low temperature. True or False: the mechanism of the Diels-Alder reaction is three π bonds break; one σ bond and two π bonds form.

